
ORGANIC CHEM.-S.M.+STD.GDE(LL)-W/ACCESS
8th Edition
ISBN: 9780134777658
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13.6, Problem 17P
Using curved arrows, show the principal fragments you would expect to sec in the mass spectrum of each of the following compounds:
- a. CH3CH2CH2CH2CH2OH
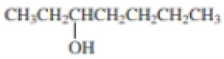
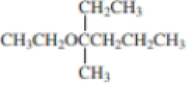
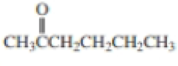
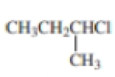
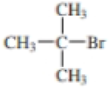
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify two peaks that are expected to appear in the mass spectrum of 3-pentanol. For each peak, identify the fragment associated with the peak.
Using curved arrows, show the principal fragments you would expect to see in the mass spectrum of each of the following compounds:
Identify the fragments in this mass spectrum of hexanoic acid.
Chapter 13 Solutions
ORGANIC CHEM.-S.M.+STD.GDE(LL)-W/ACCESS
Ch. 13.1 - Which of the following fragments produced in a...Ch. 13.2 - What distinguishes the mass spectrum of...Ch. 13.2 - What is the most likely m/z value for the base...Ch. 13.3 - Prob. 5PCh. 13.3 - a. Suggest possible molecular formulas for a...Ch. 13.3 - If a compound has a molecular ion with an...Ch. 13.3 - Identify the hydrocarbon that has a molecular ion...Ch. 13.4 - Predict the relative intensities of the molecular...Ch. 13.5 - Which molecular formula has an exact molecular...Ch. 13.5 - Prob. 11P
Ch. 13.6 - Sketch the mass spectrum expected for...Ch. 13.6 - The mass spectra of 1-methoxybutane,...Ch. 13.6 - Primary alcohols have a strong peak at m/z = 31....Ch. 13.6 - Identify the ketones responsible for the mass...Ch. 13.6 - Prob. 16PCh. 13.6 - Using curved arrows, show the principal fragments...Ch. 13.6 - The reaction of (Z)-2-pentene with water and a...Ch. 13.9 - a. Which is higher in energy: electromagnetic...Ch. 13.9 - Prob. 20PCh. 13.13 - Prob. 21PCh. 13.14 - Which occur at a larger wavenumber: a. the C O...Ch. 13.14 - Prob. 23PCh. 13.14 - Prob. 24PCh. 13.14 - Rank the following compounds from highest...Ch. 13.14 - Which shows an O H stretch at a larger...Ch. 13.16 - Prob. 27PCh. 13.16 - a. An oxygen-containing compound shows an...Ch. 13.16 - Prob. 29PCh. 13.16 - For each of the following pair of compounds, name...Ch. 13.17 - Which of the following compounds has a vibration...Ch. 13.17 - Prob. 32PCh. 13.18 - A compound with molecular formula C4H6O gives the...Ch. 13.20 - Prob. 34PCh. 13.20 - Prob. 35PCh. 13.21 - Predict the max of the following compound:Ch. 13.21 - Prob. 37PCh. 13.23 - a. At pH = 7 one of the ions shown here is purple...Ch. 13.23 - Prob. 39PCh. 13.23 - Prob. 40PCh. 13 - In the mass spectrum of the following compounds,...Ch. 13 - Prob. 42PCh. 13 - Draw structures for a saturated hydrocarbon that...Ch. 13 - Rank the following compounds in order of...Ch. 13 - For each of the following pairs of compounds,...Ch. 13 - a. How could you use IR spectroscopy to determine...Ch. 13 - Assuming that the force constant is approximately...Ch. 13 - Norlutin and Enovid are ketones that suppress so...Ch. 13 - In the following boxes, list the types of bonds...Ch. 13 - A mass spectrum shows significant peaks at m/z. =...Ch. 13 - Prob. 51PCh. 13 - Prob. 52PCh. 13 - Prob. 53PCh. 13 - The IR spectrum of a compound with molecular...Ch. 13 - Rank the following compounds from highest...Ch. 13 - Rank the following compounds from highest...Ch. 13 - What peaks in their mass spectra can be used to...Ch. 13 - Prob. 58PCh. 13 - Which one of the following five compounds produced...Ch. 13 - Prob. 60PCh. 13 - Each of the IR spectra shown below is accompanied...Ch. 13 - Prob. 62PCh. 13 - Prob. 63PCh. 13 - How can IR spectroscopy distinguish between...Ch. 13 - Prob. 65PCh. 13 - Prob. 66PCh. 13 - Give approximate wavenumbers for the major...Ch. 13 - Prob. 68PCh. 13 - Which one of the following live compounds produced...Ch. 13 - Phenolphthalein is an acid-base indicator. In...Ch. 13 - Prob. 71PCh. 13 - How can you use UV spectroscopy to distinguish...Ch. 13 - Prob. 73PCh. 13 - The IR and mass spectra for three different...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Predict the masses and the structures of the most abundant fragments observed in the mass spectra of the followingcompounds.(a) 2-methylpentane (b) 3-methylhex-2-ene (c) 4-methylpentan-2-ol(d) 2-methyl-1-phenylpropanearrow_forwardIn the mass spectrum of the following compounds, which is the tallest—the peak at m/z = 57 or the peak at m/z = 71? a. 3-methylpentane b. 2-methylpentanearrow_forwardIdentify the expected base peak in the mass spectrum of 2,2,3-trimethylbutane. Draw the fragment associated with this peak and explain why the base peak results from this fragment.arrow_forward
- Identify the key fragment structures of 4'-Bromoacetophenone based on the peaks of its attached mass spectrum.arrow_forwardWhich amomg the fragments below will be dectected by mass Spectrophotometer? [CH3CH3]+ CH3CH3 •CH2CH3 [CH3CH4]- i and iii only ii ii and iv only i only iiarrow_forwardIdentify the fragments in this mass spectrum of 2,2-dimethylbutanearrow_forward
- Using the Molecular Ion to Identify a Compound Pent-1-ene and pent-1-yne are low-boiling hydrocarbons that have different molecular ions in their mass spectra. Match each hydrocarbon to its mass spectrum.arrow_forwardWhich amomg the fragments below will be dectected by mass Spectrophotometer? [CH3CH3]+ CH3CH3 •CH2CH3 [CH3CH4]-arrow_forwardLabel each of the attached in the mass spectrum of hexan-2-ol[CH3CH(OH)CH2CH2CH2CH3]: the molecular ion, the base peak, thefragment resulting from the loss of H2O, and α cleavage fragmentsarrow_forward
- Draw all four resonance forms of the fragment at m>z 73 in the mass spectrum of pentanoic acidarrow_forwardFrom the given IR and mass spectra of the unknown compound: 1. Which element is surely present in the compound? a. F b. O c. Br d. N 2. Which statement is true? a. It has no saturation b. It is an acid anhydride c. It contains -CH2- next to benzene ring d. All are falsearrow_forwardIdentifying Isomers from Fragmentation Patterns Which mass spectrum (A or B) corresponds to each isomer: 2methylpentane [(CH3)2CHCH2CH2CH3] or 2,2-dimethylbutane [(CH3)3CCH2CH3]?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY