
Concept explainers
Platinum nanoparticles of diameter ~2 nm are important catalysts in carbon monoxide oxidation to carbon dioxide. Platinum crystallizes in a face-centered cubic arrangement with an edge length of 3.924Å. (a) Estimate how many platinum atoms would fit into a 2.0-nm sphere; the volume of a sphere is (4/3). 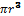
 Recall that 1Å= 1×10-10 m and 1nm = 1×10-9 m. (b) Estimate how many platinum atoms are on the surface of a 2.0-nm Pt sphere, using the surface area of a sphere (4
Recall that 1Å= 1×10-10 m and 1nm = 1×10-9 m. (b) Estimate how many platinum atoms are on the surface of a 2.0-nm Pt sphere, using the surface area of a sphere (4
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
CHEMISTRY THE CENTRAL SCIENCE 14TH EDI
- A hypothetical element, Le, has a body-centered cubic structure with a density of 7.89 g/cm3. Calculate its atomic radius in pm. Atomic Mass: Le: 58.348 g/mol Round to 2 decimal placesarrow_forward4. Lithium bromide crystalizes as shown (right) where the lithium ions are represented as white circles and the bromide ions are represented as filled circles. Give all answers to four significant figures. (a) What is the mass of one unit cell in grams? (b) If the density of LiBr is 3.464 g/mL, what is the edge length of the unit cell in pm? (c) Calculate the Li-Br distance in the crystal structure in pm.arrow_forwardThe student finds the following particulate model of AgCl(s). Assuming the crystal structures are similar, how should the student modify the model to represent CuCl(s)? Justify your answer in terms of the radius and arrangement of electrons in the ions.arrow_forward
- Copper (Cu) has an fcc structure, where the mass of a Cu atom is 63 in atomic mass units.The density of Cu is 8.9×103 kgm−3.Tungsten (W) typically has a bcc structure, where the mass of a W atom is 184 in atomicmass units. The density of W is 19.3×103 kgm−3.(i) Use the above data to find the radius of a Cu atom, and of a W atom.(ii) Which atom has the larger radius, and why?You may use the fact that 1 atomic mass unit is equivalent to 1.66 × 10−27 kg.arrow_forward1) Palladium (electron configuration is [Kr]4d¹) crystallizes in a face-centered cubic unit cell. its density is 12.0 g/cm³. (NA = 6.02 x 10²³) (a) What is the total mass of palladium atoms contained within one unit cell? (b) Calculate the atomic radius (r) of Pd.arrow_forwardCopper (Cu) has an fcc structure, where the mass of a Cu atom is 63 in atomic mass units. The density of Cu is 8.9x10³ kgm-³. Tungsten (W) typically has a bcc structure, where the mass of a W atom is 184 in atomic mass units. The density of W is 19.3x10³ kgm-³. (i) Use the above data to find the radius of a Cu atom, and of a W atom. (ii) Which atom has the larger radius, and why? You may use the fact that 1 atomic mass unit is equivalent to 1.66 × 10-27 kg.arrow_forward
- The ionic compound, CsI, forms a body centered cubic structure (see below) where the anions occupy the corners of the crystal (red) and the cations the body centered position (green). If the density of the compound is 4.51 g/cm^3 and the ionic radius of Cs + is 169 pm, what is the ionic radius of I − in pm? Key Concept: The atoms are touching the diagonal in the center of the cube. This makes it equal to the sum of 2 times the radius of each atom. Strategy: Determine the length of the diagonal,d, and recall that the length of the side, l, is given by l = d/3 0.5. This is a result of the use of the Pythagorean theorem relating the length of the side to d.With the length of the side you can determine the volume of the unit cell.From the number of atoms in the unit cell determine the mass of the unit cell.Use these to get the density.arrow_forwardNickel exhibits a cubic crystal structure with a lattice constant (a) value of 0.353 nm. It has an atomic weight of 58.70 g/mol and a density of 8.83 g/cm3. (a) What is the crystal structure of Ni? (b) Determine its atomic radius in nm. Present your answer in three (3) decimal places.arrow_forwardA hypothetical element, Le, has a body centered cubic structure with a density of 6.93 g/cm3. Calculate its atomic radius in pm. Atomic Mass: Le: 55.963 g/molarrow_forward
- Tantalum is a rare, hard, blue-gray, lustrous transition metal that is highly corrosion-resistant. It has a density of 17.6 grams per cubic centimeter and a molecular weight of 185.23 g/mol. What is the atomic radius of Ta if it adopts the body centered cubic unit structure? Express answer in scientific notation (e.g. 3.47e-23).arrow_forwardQ619 Calculate the radius of a copper atom. Copper has a face centered cubic (FCC) crystal structure with a volume density of 8.95 x 106g m-3 and an atomic weight of 63.54 g mol-1 (Given the Avogadro’s, NA = 6.023 x 1023 atoms mol-1arrow_forward6.023 x 1023 Magnesium (Mg) has an HCP crystal structure and a density of 1.740 g/cm³. What is the volume of its unit cell in cubic meters? (Given: Atomic weight of Mg is 24.31 g/mole and N₁ atoms/mole) (a) 9.28 x 10-23 m³. (b) 9.28 x 10-26 m³ (c) 1.39 x 10-25 m³ (d) 1.39 x 10-28 m³ 2.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

