
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
9th Edition
ISBN: 9781292151229
Author: Wade, LeRoy G.
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 14.49SP
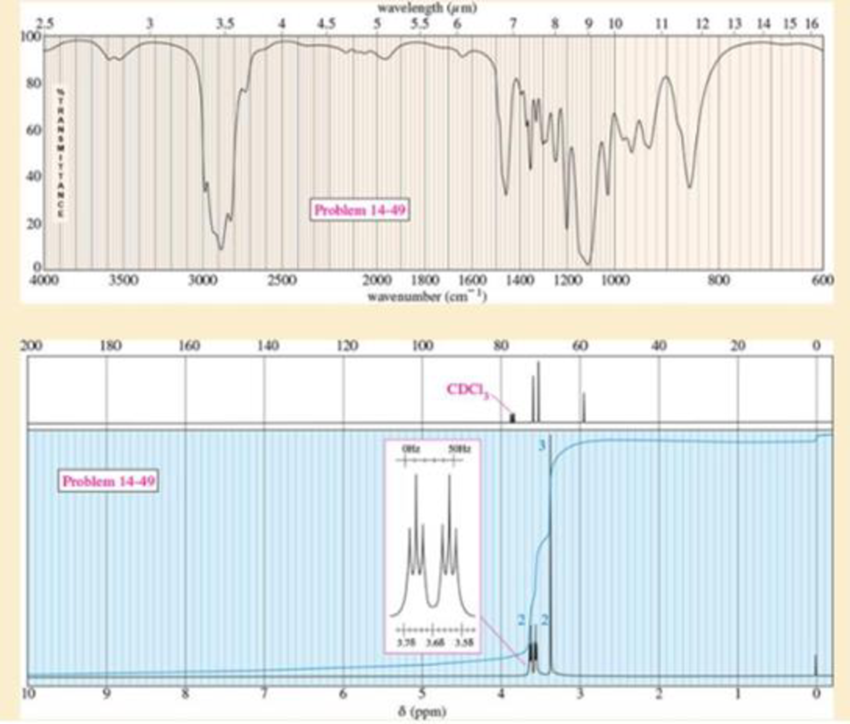
An acid-catalyzed reaction was carried out using methyl cellosolve (2-methoxyethanol) as the solvent. When the 2-methoxyethanol was redistilled, a higher-boiling fraction (bp 162 °C) was also recovered. The mass spectrum of this fraction showed the molecular weight to be 134. The IR and NMR spectra are shown here. Determine the structure of this compound and propose a mechanism for its formation.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Provide reaction mechanisms for the following transformations
Choose the right reagent or series of reagents from the ones listed below to prepare 2-pentanone from acetylene.
NaNH2NaNH2 followed by CH3CH2CH2BrCH3CH2CH2Br, then disiamylborane followed by H2O2H2O2, HO−HO−
H2H2, Lindlar followed by H2OH2O, H+H+
NaNH2NaNH2 and CH3CH2CHOCH3CH2CHO
NaNH2NaNH2 followed by H2OH2O, H+H+
NaNH2NaNH2 followed by CH3CH2CH2BrCH3CH2CH2Br, then H2OH2O, H2SO4H2SO4, HgSO4
Choose one.
Show the major organic product and mechanism for the transformations below.
Chapter 14 Solutions
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
Ch. 14.2C - Rank the given solvents in decreasing order of...Ch. 14.2D - Prob. 14.2PCh. 14.2D - In the presence of 18-crown-6, potassium...Ch. 14.3B - Give a common name (when possible) and a...Ch. 14.3C - Prob. 14.5PCh. 14.3C - Name the following heterocyclic ethers.Ch. 14.4 - Propose a fragmentation to account for each...Ch. 14.5 - Prob. 14.8PCh. 14.5 - Prob. 14.9PCh. 14.6 - Prob. 14.10P
Ch. 14.7 - Explain why bimolecular condensation is a poor...Ch. 14.7 - Prob. 14.12PCh. 14.7 - Prob. 14.13PCh. 14.8 - Prob. 14.14PCh. 14.8 - Prob. 14.15PCh. 14.8 - Prob. 14.16PCh. 14.10A - Prob. 14.17PCh. 14.10A - Prob. 14.18PCh. 14.10B - Prob. 14.19PCh. 14.11B - Show how you would accomplish the following...Ch. 14.11B - Prob. 14.21PCh. 14.12 - Prob. 14.22PCh. 14.12 - Prob. 14.23PCh. 14.12 - Prob. 14.24PCh. 14.13 - Prob. 14.25PCh. 14.13 - Prob. 14.26PCh. 14.14 - Prob. 14.27PCh. 14.15 - Give the expected products of the following...Ch. 14 - Write structural formulas for the following...Ch. 14 - Give common names for the following compounds. a....Ch. 14 - Give IUPAC names for the following compounds. a....Ch. 14 - Glycerol (propane-1,2,3-triol) is a viscous syrup...Ch. 14 - Prob. 14.33SPCh. 14 - Show how you would make the following ethers,...Ch. 14 - (A true story.) An inexperienced graduate student...Ch. 14 - Prob. 14.36SPCh. 14 - a. Show how you would synthesize the pure (R)...Ch. 14 - a. Predict the values of m/z and the structures of...Ch. 14 - The following reaction resembles the...Ch. 14 - Prob. 14.40SPCh. 14 - Prob. 14.41SPCh. 14 - Prob. 14.42SPCh. 14 - Give the structures of the intermediates...Ch. 14 - Prob. 14.44SPCh. 14 - Show how you would synthesize the following ethers...Ch. 14 - Prob. 14.46SPCh. 14 - Prob. 14.47SPCh. 14 - Prob. 14.48SPCh. 14 - An acid-catalyzed reaction was carried out using...Ch. 14 - Propylene oxide is a chiral molecule. Hydrolysis...Ch. 14 - Prob. 14.51SPCh. 14 - Prob. 14.52SPCh. 14 - Prob. 14.53SPCh. 14 - Prob. 14.54SPCh. 14 - In 2012, a group led by Professor Masayuki Satake...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Knoevenagel condensation is a reaction involving an active methylene compound (a CH2 flanked by two electron-withdrawing groups) and an aldehyde and ketone. Propose a mechanism for the reaction below.arrow_forwardThe tosylate of a primary alcohol normally undergoes an SN2 reaction with hydroxide ion to give a primary alcohol. Reaction of this tosylate, however, gives a compound of molecular formula C7H12O. Propose a structural formula for this compound and a mechanism for its formation.arrow_forwardPropose a mechanism to account for the formation of 3, 5-dimethyl- pyrazole from hydrazine and 2, 4-pentanedione. Look carefully to see what has happened to each carbonyl carbon in going from starting material to product.arrow_forward
- Solvolysis of the following bicyclic compound in acetic acid gives a mixture of products, two of which are shown. The leaving group is the anion of a sulfonic acid, ArSO3H. A sulfonic acid is a strong acid, and its anion, ArSO3, is a weak base and a good leaving group. Propose a mechanism for this reaction.arrow_forwardEthylene oxide is the starting material for the synthesis of 1,4-dioxane. Propose a mechanism for each step in this synthesis.arrow_forwardShow the products you would obtain by acid-catalyzed reaction of cyclohexanone with ethylamine, CH3CH2NH2, and with diethylamine, (CH3CH2) 2NH.arrow_forward
- Provide the reagents and mechanism for each step of the synthesis.arrow_forwardThe reaction of 2,2-dimethyl-1-propanol with HBr is very slow and forms 2-bromo-2-methylbutane as the main product Propose a mechanism to explain these observationsarrow_forwardStep by step reaction of 2-methyl propene with methanol in acid-catalyzed medium showing the mechanism, show the product to be formedarrow_forward
- Propose a mechanism for the acid-catalyzed hydration of 1-methylcyclohexene to give 1-methylcyclohexanol. Which step in your mechanism is rate-determining?arrow_forwardProvide the mechanism for the following transformation. D = 2Harrow_forward3. Provide a detailed, stepwise mechanism for the acid-catalyzed reaction of 2-butanone with ethylene glycol (HOCH2CH2OH) to produce an acetal.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License