
(a)
Interpretation:
The achiral molecule should be identified.
Concept introduction:
Chiral:
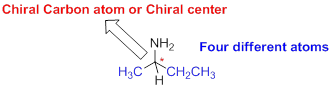
The carbon atom which attached to the four different atoms is called as chiral carbon. A molecule is non superimposable on its mirror image is called chiral molecule. The molecule is called as chiral molecule, the carbon is called as chiral carbon.
Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
(b)
Interpretation:
The achiral molecule should be identified.
Concept introduction:
Chiral:
The carbon atom which attached to the four different atoms is called as chiral carbon. A molecule is non superimposable on its mirror image is called chiral molecule. The molecule is called as chiral molecule, the carbon is called as chiral carbon.

Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
(c)
Interpretation:
The achiral molecule should be identified.
Concept introduction:
Chiral:
The carbon atom which attached to the four different atoms is called as chiral carbon. A molecule is non superimposable on its mirror image is called chiral molecule. The molecule is called as chiral molecule, the carbon is called as chiral carbon.

Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
(d)
Interpretation:
The achiral molecule should be identified.
Concept introduction:
Chiral:
The carbon atom which attached to the four different atoms is called as chiral carbon. A molecule is non superimposable on its mirror image is called chiral molecule. The molecule is called as chiral molecule, the carbon is called as chiral carbon.

Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
(e)
Interpretation:
The achiral molecule should be identified.
Concept introduction:
Chiral:
The carbon atom which attached to the four different atoms is called as chiral carbon. A molecule is non superimposable on its mirror image is called chiral molecule. The molecule is called as chiral molecule, the carbon is called as chiral carbon.

Achiral:
A molecule is superimposable on its mirror image is called achiral molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
EP FUND.OF GENERAL,ORG...-MOD.MASTERING
- 11.68 Acetylene (ethyne) gas reacts with oxygen and burns at 3300 °C in an acetylene torch. (7.4, 7.7, 8.6, 11.4, 11.5) a. Write the balanced chemical equation for the complete combustion of acetylene. b. What is the molar mass of acetylene? c. How many grams of oxygen are needed to react with 8.5 L of acetylene gas at STP? d. How many liters of CO2 gas at STP are produced when 30.0 g of acetylene undergoes combustion?arrow_forwardWhat is the IUPAC name of the following compound? (No need to provide E/Z designation.) CO₂Harrow_forwardWrite the formulas of potassium salicylate and disodium oxalate (refer to Table 17.1).arrow_forward
- A prescription came in for 34% salicylic acid. The pharmacy has 95% salicylic and 10%salicylic acid available. Write a recipe for 500mg of 34% salicylic acid.arrow_forwardThe empirical formula of the sugar glucose is C6H12O6. (a) How many moles are there in 270 g of glucose? (b) Calculate the molarity of a solution of 324 g of glucose dissolved in 2.0 l of water.arrow_forwardThe following compound can be described as a(an)arrow_forward
- What is the ionisable group of phenazopyridine? With illustrationsarrow_forwardEach of the following materials has an ester that is responsible for its smell and/or flavor. Search the internet and determine what that ester is, draw its structure, and what carboxylic acid and alcohol are used to form it.(a) Juicy Fruit gum flavoring(b) Peach odor(c) Apple odor(d) Rum odorarrow_forwardDraw structural formulas for all of the following. Q.) Alcohols with the molecular formula C4H10Oarrow_forward
- Balance the following equation, and tell how many moles of nickel will reactwith 9.81 mol of hydrochloric acid.arrow_forwardThe following data was collected for n-pentane and isopentane: Compound P, (kPa) n-pentane 282.9 at 70°C 895.5 at 120°C Isopentane 955.9 at 115°C 1152.4 at 125°C а) Develop a correlation for the vapor pressure of Isopentane as a function of temperature. Clearly state the units. b) Calculate the vapor pressure of Isopentane at 120°C. c) Estimate the heat of vaporization of Isopentane by using the data in the table. A closed container contains a mixture of 65.0 mol% n-pentane and 35.0 mol% isopentane. The system is initially all liquid at 120.0°C and high pressure and the pressure is gradually reduced at a constant temperature. d) i. Calculate the pressure (in kPa) at which the first bubble of vapor forms. ii. What is the composition (in mole fractions) of the first bubble of vapor at the conditions found in part (i)? A new mixture contains cyclohexane (Pv=288.3kPa at 120°C), n-pentane and isopentane at 120°C and high pressure. The pressure is reduced at constant temperature to a point…arrow_forwardWhen pentane is exposed to Br2 in the presence of light, a halogenation reaction occurs. Write the formulas of:(a) All possible products containing only one bromine(b) All possible products containing two bromines that are not on the same carbonarrow_forward
