
Concept explainers
(a)
Interpretation:
The alcohol should be identified as primary, secondary or tertiary alcohol.
Concept introduction:
Classification of alcohol:
Alcohols can be classified in to three types as
Primary alcohol,
Secondary alcohol,
Tertiary alcohol
Primary alcohol:
Hydroxyl group (
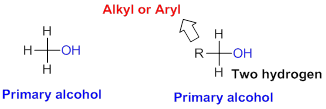
Secondary alcohol:
Hydroxyl group (
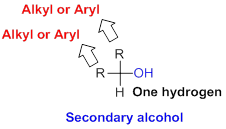
Tertiary alcohol:
Hydroxyl group (
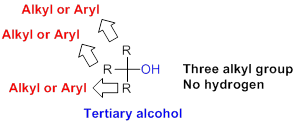
(b)
Interpretation:
The hydrophilic or hydrophobic part and solubility property in water should be identified for the given compounds.
Concept introduction:
Alcohols have two different parts
- (1) Hydrophilic (water-loving),
- (2) Hydrophobic (water-fearing).
Hydrophilic (water-loving):
The compound has the hydroxyl group (

Hydrophobic (water-fearing):
The compound has alkyl chain group is called as hydrophilic.
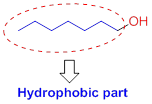
Solubility of the alcohol in water:
The solubility of the alcohol is mainly depends on the hydrophilic part as well as length of the alkyl chain. If the compound has more hydroxyl group or less number of carbon chain which is soluble in water. If the compound has hydroxyl group with more number of carbon which is insoluble in water because of hydrophobic part.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
EP FUND.OF GENERAL,ORG...-MOD.MASTERING
- A prescription came in for 34% salicylic acid. The pharmacy has 95% salicylic and 10%salicylic acid available. Write a recipe for 500mg of 34% salicylic acid.arrow_forwardThe concentration of alcohol 1CH3CH2OH2 in blood, calledthe “blood alcohol concentration” or BAC, is given in unitsof grams of alcohol per 100 mL of blood. The legal definitionof intoxication, in many states of the United States, is that theBAC is 0.08 or higher. What is the concentration of alcohol,in terms of molarity, in blood if the BAC is 0.08?arrow_forwardWrite the formulas of potassium salicylate and disodium oxalate (refer to Table 17.1).arrow_forward
- The following compound can be described as a(an)arrow_forwardYou have a 0.5M stock solution of NaCl (formula weight: 58.4 g/mole), a 0.15M stock solution of glucose (formula weight: 180.2g/mole), and a bottle of solid Tris base (formula weight: 121.1g/mole). How would you prepare (be specific) 25 mL of a single solution containing 10mM Tris, 15mM glucose, and 5mM NaCl.arrow_forwardYou are asked to prepare 50 ml of a 1:100 rifampin (rifadin) and you have in stock a 1:20 rifampin suspension. How many milliliter of the 1:20 suspension and how many milliliter of water will you need?arrow_forward
- A patient brings in a prescription for their child that asks for Amoxicillin 250mg/5mL. The directions say, "2 tsp po bid." Your pharmacy is low on inventory and only has 80mL of Amoxicillin 125mg/5mL (Add 65 mL of water to powder). How many mL of water should you add to get a concentration of 250mg/5mL?arrow_forwardDraw the alcohol used to produce each of the following ketones. Include all hydrogen atoms.arrow_forwardThe explosive trinitrotoluene (TNT) is made by carrying out three successive nitration reactions on toluene. If these nitrations only occur in the ortho and para positions relative to the methyl group, what is the structure of TNT?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





