
Concept explainers
(a)
Interpretation:
The specific bonds in the given molecules and the wavenumber according to their absorption in given range have to be identified.
Concept introduction:
IR spectral studies: It is a spectroscopic technique which is used to determine the functional groups present in the given compound sample by absorbing characteristic frequency in particular range with respect to the group present in the given sample.
(a)
Explanation of Solution
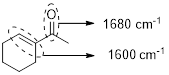
The structure shows that the molecule contains one carbonyl group and one carbon-carbon double bond. Analyzing the structure shows that the carbonyl group and the double bonds are conjugated hence, the carbonyl group shows signal near to 1680
(b)
Interpretation:
The specific bonds in the given molecules and the wavenumber according to their absorption in given range have to be identified.
Concept introduction:
IR spectral studies: It is a spectroscopic technique which is used to determine the functional groups present in the given compound sample by absorbing characteristic frequency in particular range with respect to the group present in the given sample.
(b)
Explanation of Solution
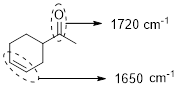
The structure shows that the molecule contains one carbonyl group and one carbon-carbon double bond. Analyzing the structure shows that the carbonyl group and the double bond are not conjugated they are isolated hence the carbonyl group produce signal near 1720
(c)
Interpretation:
The specific bonds in the given molecules and the wavenumber according to their absorption in given range have to be identified.
Concept introduction:
IR spectral studies: It is a spectroscopic technique which is used to determine the functional groups present in the given compound sample by absorbing characteristic frequency in particular range with respect to the group present in the given sample.
(c)
Explanation of Solution
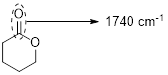
The structure shows that the molecule contains one ester group. Therefore, the carbonyl group presents in the ester exhibit a signal near to 1740
(d)
Interpretation:
The specific bonds in the given molecules and the wavenumber according to their absorption in given range have to be identified.
Concept introduction:
IR spectral studies: It is a spectroscopic technique which is used to determine the functional groups present in the given compound sample by absorbing characteristic frequency in particular range with respect to the group present in the given sample.
(d)
Explanation of Solution
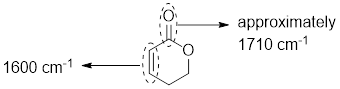
The structure shows that the molecule contains one ester group and one carbon-carbon double bond where both are conjugated hence carbonyl group of ester produce signal near 1710
Want to see more full solutions like this?
Chapter 14 Solutions
ORGANIC CHEMISTRY (LL) >CUSTOM PACKAGE<
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





