
Concept explainers
Identify the
(a) Isomer A has the
(b) Isomer B has three peaks in its
(c) Isomer C has two peaks in its
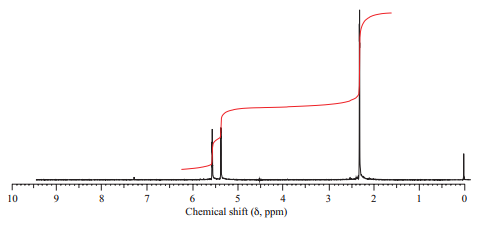
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
ORGANIC CHEMISTRY-W/STUD.SOLN.MAN.
- A compound, whose formula is C7H12, is known to have a six-membered ring. In its UV–vis spectrum, the longest-wavelength λmax appears at 191 nm. Draw four isomers that are consistent with these results.arrow_forwardThe following 1H NMR spectra are for four compounds, each with molecular formula of C6H12O2. Identify the compounds.arrow_forwardThe two compounds 1,2-dibromobenzene and 1 3-dibromobenzene both have the molecular formula C6H4Brz. a)Draw the chemical structures of the two molecules. b)Can these two molecules be distinguished by 1H NMR spectroscopy? Give reasons for your answer and describe the number of proton environments and their splitting for both molecules. Can these two molecules be distinguished by 13C NMR spectroscopy? Give reasons for your answer. Can these two molecules be distinguished by IR spectroscopy? Give reasons for your answer and describe the main IR signals (with frequencies) you expect to see for both molecules. Can these two molecules be distinguished by mass spectrometry? Give reasons for your answer.arrow_forward
- What wavelength is the peak absorbance of CoCl2?arrow_forwardPredict the theoretical number of different NMR signals produced by each compound, and give approximate chemical shifts. Point out any diastereotopic relationships. (a) Ph¬CHBr¬CH2Br (b) vinyl chloridearrow_forwardWhat is the index of hydrogen deficiency of a compound with the empirical formula C5H11NO? a) 0 b) 1 c) 2 d) 3arrow_forward
- Analyze the 1H AND 13C NMR spectrum of C8H9NO given below and draw the structure of the compound.arrow_forwardWhat does the (2H,ddd, J=8.3 Hz) mean? What would be the possible structure of this? The compound is C8H8O2arrow_forwardPropose a structural formula for the analgesic phenacetin, molecular formula C10H13NO2, based on its 1H-NMR spectrum.arrow_forward
- what is the constitutional isomer for this spectrum? (C5h12O)arrow_forwardCompound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
