
ORGANIC CHEMISTRY-EBOOK>I<
9th Edition
ISBN: 9781305084414
Author: McMurry
Publisher: INTER CENG
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14.2, Problem 4P
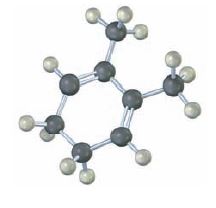
Give the structures of both 1, 2 and 1, 4 adducts resulting from reaction of 1 equivalent of HBr with the following compound:
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
p-toluic acid is prepared from 3.42 grams of p-Bromotoluene. When p-Bromotoluene was mixed with dry ether, the mixture was warmed until the ether begins to boil (34.6 °C). When the reaction is almost complete, dry ice (3-4 mL in a beaker) was prepared and the mixture was poured into it. After some time, 5 mL of 6 N HCl was added. The solvent used for the recrystallization is 100 mL of 30% ethanol. The reaction equation is given on the figure.
What is the actual yield?
What is the theoretical yield?
How many moles of p-toluic acid was produced?
5A
Explain which of the following statements are true and which are false.
i. N,N'-dicyclohexylurea (DCU) is the dehydrating reagent that converts a
carboxylic acid to the corresponding anhydride.
ii. The fact that carbonyl compounds have a lower boiling point than
alcohols with the same C atoms is because the molecular dipole moment of
carbonyl compounds is less than that of alcohols.
iii. 3-Methylbut-2-en-2-ol is the only tautomeric structure of 3-
methylbutan-2-one.
Provide the necessary compounds to form heptane according to the given reagents.
Ether
A
2 Li B Cul
C + D
Heptane
Pentane
For your answers, use the name of the compounds instead of their formulas.
A =
B =
C=
D=
Submit your solution showing the structures of A, B, C, and D.
Chapter 14 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
Ch. 14.1 - Prob. 1PCh. 14.2 - Give the structures of both 1, 2 and 1, 4 adducts...Ch. 14.2 - Prob. 3PCh. 14.2 - Give the structures of both 1, 2 and 1, 4 adducts...Ch. 14.3 - Prob. 5PCh. 14.3 - Prob. 6PCh. 14.5 - Predict the product of the following Diels–Alder...Ch. 14.5 - Prob. 8PCh. 14.5 - Which of the following dienes have an s-cis...Ch. 14.5 - Predict the product of the following Diels–Alder...
Ch. 14.6 - Prob. 11PCh. 14.6 - Prob. 12PCh. 14.7 - Prob. 13PCh. 14.7 - Prob. 14PCh. 14.8 - Which of the following compounds would you expect...Ch. 14.SE - Prob. 16VCCh. 14.SE - Show the product of the Diels–Alder reaction of...Ch. 14.SE - Prob. 18VCCh. 14.SE - Prob. 19VCCh. 14.SE - Prob. 20MPCh. 14.SE - Prob. 21MPCh. 14.SE - In light of your answer to Problem 14-21 propose...Ch. 14.SE - Luminol, which is used by forensic scientists to...Ch. 14.SE - Prob. 24MPCh. 14.SE - Give IUPAC names for the following compounds:Ch. 14.SE - Prob. 26APCh. 14.SE - Prob. 27APCh. 14.SE - Electrophilic addition of Br2 to isoprene...Ch. 14.SE - Prob. 29APCh. 14.SE - Prob. 30APCh. 14.SE - Predict the products of the following...Ch. 14.SE - 2,3-Di-tert-butyl-1,3-butadiene does not undergo...Ch. 14.SE - Prob. 33APCh. 14.SE - Prob. 34APCh. 14.SE - Prob. 35APCh. 14.SE - Prob. 36APCh. 14.SE - Rank the following dienophiles in order of their...Ch. 14.SE - Prob. 38APCh. 14.SE - Prob. 39APCh. 14.SE - Prob. 40APCh. 14.SE - Although the Diels–Alder reaction generally...Ch. 14.SE - Prob. 42APCh. 14.SE - Tires whose sidewalls are made of natural rubber...Ch. 14.SE - Prob. 44APCh. 14.SE - Prob. 45APCh. 14.SE - Prob. 46APCh. 14.SE - Would you expect allene, H2C = C = CH2, to show a...Ch. 14.SE - The following ultraviolet absorption maxima have...Ch. 14.SE - Prob. 49APCh. 14.SE - -Ocimene is a pleasant-smelling hydrocarbon found...Ch. 14.SE - Draw the resonance forms that result when the...Ch. 14.SE - Prob. 52APCh. 14.SE - Treatment of 3,4-dibromohexane with strong base...Ch. 14.SE - Prob. 54APCh. 14.SE - Prob. 55APCh. 14.SE - Prob. 56APCh. 14.SE - Prob. 57APCh. 14.SE - Prob. 58APCh. 14.SE - Hydrocarbon A, C10H14, has a UV absorption at...Ch. 14.SE - Prob. 60APCh. 14.SE - Prob. 61APCh. 14.SE - Prob. 62APCh. 14.SE - Prob. 63APCh. 14.SE - Prob. 64APCh. 14.SE - The double bond of an enamine (alkene + amine) is...Ch. 14.SE - Prob. 66AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- When 2-pentene is treated with Cl2 in methanol, three products are formed. Account for the formation of each product (you need not explain their relative percentages).arrow_forward1. In the reactions involving the three isomeric alcohols with the formula C4H9OH, describewhat each of the following tests showed about reactivity of the -OH group and reactions of 1°,2°, and 3° alcohols.• the test with neutral KMnO4• the test with concentrated HCl2. Predict how the fourth alcohol with the formula C4H10O would react if tested with:• 0.01 M KMnO4• concentrated HCl at room temperatureExtend FurtherUse your observations of the solutions formed in the previous experiments and yourunderstanding of alcohols to complete a table like the one shown below. Research the meltingand boiling points to verify your answers.arrow_forwardGive the major organic product(s) for the following reaction Br₂ (1 mole) ? room temperaturearrow_forward
- 2. What kind of results do you expect to see when the following compounds are mixed together with the given test solution? Indicate positive or negative in the table below. (a) O with 2,4-dinitrophenylhydrazine (b) with Benedict's reagent (c) with iodoform reagent (d) with Tollen's reagent H JH H CH3arrow_forwardWrite reactions of 2-methyl propane with the following reagents: a. Cl2/t °C;arrow_forwardMaterials • 1-butanol • 2-butanol • 2-methyl-2-propanol • 3 mol/L H2SO4 (aq) • 3 mol/L NaOH • 0.01 mol/L KMNO4 • concentrated (12 mol/L) HCl (aq) for demonstration use only • distilled water • 10 mL graduated cylinders or graduated medicine droppers • test tubes • test-tube rack Procedure Part 1: The comparison of three isomeric alcohols reactions with potassium permanganate 1. Construct an observation table with the following headings. Alcohol Observations at 1 minute Observations at 5 mins 1-Butanol 2-Butanol 2-Methylpropanolarrow_forward
- Reduction of ketones: How to calculate the theoretical yield in reduction of 4-tert-butylcyclohexanone with sodium borohydride? I am confused on which is the limiting reagent. we used 0.102 g of 4-t-butylcyclohexanone and 0.012g of NaBH4.arrow_forwardProvide the major product for the following reaction? (1) BH3, ether (2) H2O2, OHarrow_forwardb) Compound E with a molecular formula of CaH30 gives a positive iodoform test. он NaBH4 H3C. E CH3 i) Suggest the structure of compound E. ii) Propose an equation for the reaction between compound E and the iodoform test reagent.arrow_forward
- 19. A compound with molecular formula C6H12O2 exhibits two singlets in its 1H NMR spectrum, at d 1.4 (9H) and d 2.0 (3H). Its IR spectrum shows a strong absorption band near 1740 cm-1. What is the structure for this compound? Show the correlation of the spectra with the molecular structure.arrow_forwardGive the products formed when Benzaldehyde and Benzoic Acid are treated with the given reagents. g. LiAlH4 then H2O, H+h. 2 moles CH3OH, H+i. CH3MgCl, then H2O, H+arrow_forwardGive the major products (structure and name) of the major organic products you would expect from the reaction of m-methylaniline with the following reagents: Br2 (1 equivalent) CH3l (excess)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Seven Name Reactions in One - Palladium Catalysed Reaction (047 - 053); Author: Rasayan Academy - Jagriti Sharma;https://www.youtube.com/watch?v=5HEKTpDFkqI;License: Standard YouTube License, CC-BY