
Concept explainers
Interpretation:
The structures of all possible adducts obtainable when the diene given reacts with one equivalent of HCl are to be given.
Concept introduction:
Conjugated dienes undergo electrophilic addition reactions through the formation of an allyl carbocation. The allyl cation is resonance stabilized and the attack of chloride ion on each of these forms leads to the formation of a mixture of 1, 2- and 1, 4-addition products.
To give:
The structures of all possible adducts obtainable when the diene given reacts with one equivalent of HCl.
Answer to Problem 16VC
The compound given is 1-vinylcyclopentene.The structures of all the possible adducts obtained when it reacts with one equivalent of HCl are given below.
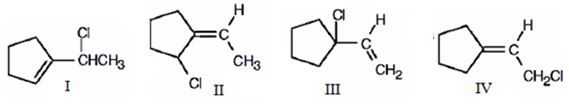
Explanation of Solution
1-Vinylcyclopentene is an unsymmetrical conjugated diene. Protonation of the CH2 in double bond and that in the ring lead to the formation of two different allyl carbocations, each of which is in resonance with another form. Hence four carbocations as shown are possible. Attack of the chloride ion on each of these carbocation leads to the formation of four different products.
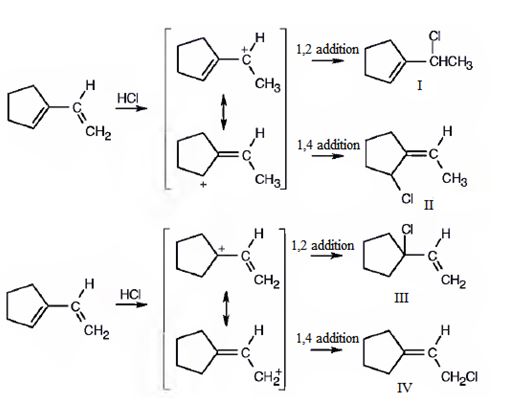
The compound given is 1-vinylcyclopentene.The structures of all the possible adducts obtained, when it reacts with one equivalent of HCl, are given below.
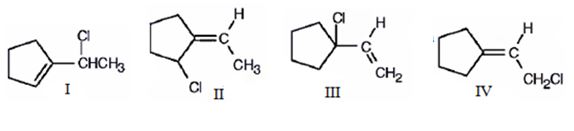
Want to see more full solutions like this?
Chapter 14 Solutions
Organic Chemistry - With Access (Custom)
- In an electrophilic addition reaction, conjugated dienes give more than one expected product. Show the products when using the reagent 1,3 –butadiene and HBr.arrow_forwardWhat two sets of a conjugated diene and a dienophile could be used to prepare the following compound?arrow_forwardShow the product of the Diels–Alder reaction of the following diene with 3-buten-2-one, H2C = CHCOCH3. Make sure you show the full stereochemistry of the reaction product.arrow_forward
- For a diels-alder reaction between anthracene and maleic anhydride, are the exo and endo forms of product 9,10-dihydroanthracene-9,10-ɑ,β-succinic acid anhydride different stereoisomers or are they the same molecule?arrow_forwardIn the beginning of this video https://www.youtube.com/watch?v=9Ng6Zv9oLzk, what reason is given on why the dienophile likes the diene?arrow_forwardShow that the [6 + 2] cyclization of hexa-1,3,5-triene with maleic anhydride is thermally forbidden but photochemicallyallowed.(e) Show the Diels–Alder product that would actually result from heating hexa-1,3,5-triene with maleic anhydride.arrow_forward
- What is the reaction mechanism of this Diels-Alder reaction between 9-anthracenemethanol and N-methylmaleimide? It is an example of a [4+2]-cycloadditionarrow_forwardWhat represents the structure of the diene to carry out the following Diels-Alder reaction?arrow_forward1,3-pentadiene reacts with HBr. When the double bond at the 3-position reacts, the hydrogen attaches to the 4-carbon, thus forming an allylic carbocation with delocalized electrons. This carbocation has another resonance structure. Because there are equal amounts of diene and hydrogen bromide, in which molecule only one of the double bonds reacts. Which of the following correctly describes this new resonance structure? a) It has a double bond between carbons 3 and 4 b) It has a positive charge on carbon 2 c) It has a positive charge on carbon 4 d) It has a double bond between carbons 2 and 3arrow_forward
- The central carbon atom of an allene is a member of two double bonds, and ithas an interesting orbital arrangement that holds the two ends of the molecule at rightangles to each other. Draw the two enantiomers of penta-2,3-diene. A model may be helpful.arrow_forwardConsider the following 2 dienes. When treated with HBr, one of these dienes yields four products while the other diene yields only two products. Explain.arrow_forwardShow the structure of the product from the Diels-Alder reaction between 2 moles of isoprene (2-methyl-1,3-butadiene) and one mole of quinone.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

