
The dependence of the equilibrium constant of a reaction on temperature is given by the van't Hoff equation:
where C is a constant. The following table gives the equilibrium constant
KP |
138 |
5.12 |
0.436 |
0.0626 |
0.0130 |
TOO |
600 |
700 |
800 |
900 |
1000 |
(a) Determine graphically the
How does this equation support the prediction based on Le Châ�telier's principle about the shift in equilibrium with temperature? (c) The vapor pressures of water are 31.82 mmHg at
Interpretation:
The
Concept Introduction:
Achemical equilibrium is a state of the chemical reaction when the rate of forward reaction becomes equal to the rate of reverse reaction and the concentrations of the products and reactants become constant, which are known as equilibrium concentrations.
When energy is released in a reaction, the reaction is called exothermic, and the reactions in which energy is absorbed are endothermic reactions.
According to Le Chatelier’s Principle, when a system at equilibrium is subjected to any change, the system tries to undo the effect of that change by shifting its equilibrium in the desired direction.
The van't Hoff equation provides information about the temperature dependence of the equilibrium constant.
Answer to Problem 125AP
Solution:
(a)
(b)
According to Le Chatelier’s principles, increase in temperature favors the forward endothermic reaction, and decrease in temperature favors an exothermic reaction.
(c)
Explanation of Solution
a)The
The
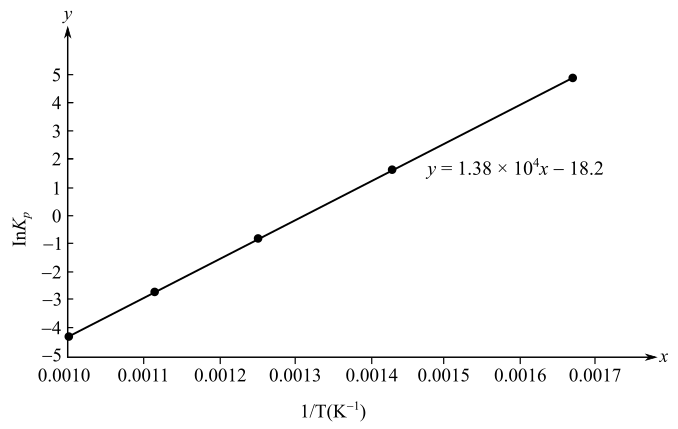
The slope of the plot is
Here,
Substitute the values of
The
b)The following equation support the prediction based on Le-Chatelier’s principle about the shift in equilibrium with temperature
The vant Hoff’s equation at two different temperatures is
Here,
From (1) and (2):
An endothermic reaction has
Then, temperature is
In a reversible reaction at equilibrium, when the equilibrium constant is more, the products are also more or the rate constant of forward reaction is more, and hence the products are more. According to LeChatelier’s principle, an increase in temperature favors endothermic reaction, and a decrease in temperature favors exothermic reaction.
c) Molar heat of vaporization of water
The vapor pressure of water is
From vant’s Hoff equation,
Here,
Substitute the values of
On further solving
The molar heat ofvaporization of water is
Want to see more full solutions like this?
Chapter 15 Solutions
Chemistry
- What is the approximate value of the equilibrium constant KP for the change C2H5OC2H5(l)C2H5OC2H5(g) at 25 C. {Vapor pressure was described in the previous Chapter on liquids and solids; refer back to this chapter to find the relevant information needed to solve this problem.)arrow_forwardThe equilibrium constant for the butane iso-butane equilibrium at 25 C is 2.50. Calculate rG at this temperature in units of kJ/mol.arrow_forwardShow that the complete chemical equation, the total ionic equation, and the net ionic equation for the reaction represented by the equation KI(aq)+I2(aq)KI3(aq) give the same expression for the reaction quotient. KI3 is composed of the ions K+ and I3-.arrow_forward
- Kc = 5.6 1012 at 500 K for the dissociation of iodine molecules to iodine atoms. I2(g) 2 I(g) A mixture has [I2] = 0.020 mol/Land [I] = 2.0 108 mol/L. Is the reaction at equilibrium (at 500 K)? If not, which way must the reaction proceed to reach equilibrium?arrow_forwardThe reaction, 3 H2(g) + N2(g) (g), has the fol lowing equilibrium constants at the temperatures given: atT=25°C,K= 2.8 X 104 at T = 500°C, A = 2.4 X IO"7 At which temperature are reactants favored? At which temperature are products favored? YVhat can you say about the reaction if the equilibrium constant is 1.2 at 127°C?arrow_forwardThe equilibrium constant Kc for the synthesis of methanol, CH3OH. CO(g)+2H2(g)CH3OH(g) is 4.3 at 250C and 1.8 at 275C. Is this reaction endothermic or exothermic?arrow_forward
- For the system SO3(g)SO2(g)+12 O2(g)at 1000 K, K=0.45. Sulfur trioxide, originally at 1.00 atm pressure, partially dissociates to SO2 and O2 at 1000 K. What is its partial pressure at equilibrium?arrow_forwardCarbon dioxide decomposes into CO and O2 at elevated temperatures. What is the equilibrium partial pressure of oxygen in a sample at 1000 C for which the initial pressure of CO2 was 1.15 atm?arrow_forwardYou place 0.600 mol of nitrogen, N2, and 1.800 mol of hydrogen, H2, into a reaction vessel at 450C and 10.0 atm. The reaction is N2(g)+3H2(g)2NH3(g) What is the composition of the equilibrium mixture if you obtain 0.048 mol of ammonia, NH3, from it?arrow_forward
- For the reaction N2(g)+3H2(g)2NH3(g) show that Kc = Kp(RT)2 Do not use the formula Kp = Kc(RT)5n given in the text. Start from the fact that Pi = [i]RT, where Pi is the partial pressure of substance i and [i] is its molar concentration. Substitute into Kc.arrow_forwardFluorine, F2, dissociates into atoms on heating. 12F2(g)F(g) The value of Kp at 842C is 7.55 102 What is the value of Kc at this temperature?arrow_forwardNitrosyl chloride, NOC1, decomposes to NO and Cl2 at high temperatures. 2 NOCl(g) ⇌ 2 NO(g) + Cl2(g) Suppose you place 2.00 mol NOC1 in a 1.00–L flask, seal it, and raise the temperature to 462 °C. When equilibrium has been established, 0.66 mol NO is present. Calculate the equilibrium constant Kc for the decomposition reaction from these data.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





