
Concept explainers
(a)
Interpretation: The
Concept introduction: Cleavage of
Answer to Problem 15.30P
The
Explanation of Solution
The energy required to break the
Thus, the increasing order of bond strength is,
The
(b)
Interpretation: The radicals resulting from the cleavage of each
Concept introduction: Primary
Answer to Problem 15.30P
The radicals resulting from the cleavage of each
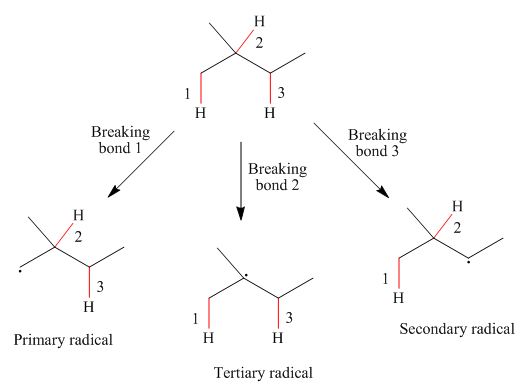
Figure 1
Explanation of Solution
Primary
The homolytic cleavage of the
The radicals resulting from the cleavage of each

Figure 1
The radicals resulting from the cleavage of each
(c)
Interpretation: The radicals resulting from the cleavage of each
Concept introduction:
Answer to Problem 15.30P
The radicals in increasing order of stability are
Explanation of Solution
The radicals resulting from the cleavage of each

Figure 1
The stability of radical depends upon the number of alkyl groups attached to the radical carbon. Therefore, stability of tertiary radical is more than secondary and primary radical. The radicals in increasing order of stability are
The radicals in increasing order of stability are
(d)
Interpretation: The
Concept introduction:
Answer to Problem 15.30P
The
Explanation of Solution
The radicals resulting from the cleavage of each

Figure 1
The stability of radical depends upon the number of alkyl groups attached to the radical carbon. Therefore, stability of tertiary radical is more than secondary and primary radical. The radicals in increasing order of stability are
Hydrogen atoms are less polarizable than alkyl groups. Therefore, alkyl group can easily donate electron density to the electron deficient carbon radical. Therefore, the increasing ease of
The
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry
- 1.) a.) Rank the molecules from 1-3 based on their rate of reactivity with NaI with 1 being the fastest. Explain the theory behind your ranking of each compound. (refer to picture 1 ) b.) Predict the reactivity of the following alkyl bromides in a substitution reaction with NaI in acetone. Rank them from 1-2 with 1 being the fastest and circle the molecule(s) that reacted in the substitution experiment? (refer to picture 2)arrow_forwardWhat is the locant of the methoxy substituent? 4-methoxy 1-methoxy 5-methoxy 3-methoxyarrow_forwardDraw all resonance structures for the carbocation formed by ortho attack of the electrophile +NO2 on attached starting material. Label any resonance structures that are especially stable or unstablearrow_forward
- How does doubling [B:] affect the rate of an E1 reaction?arrow_forwardRadical bromination of cyclohexene using NBS gives 3-bromo-1-cyclohexene. Draw the allylic radical intermediate formed during this reaction, showing both resonance structuresarrow_forward1. For the following reaction, what is the rate law ? (image) 2. is the following nucleophile strong or weak HO^- a) strong b) weak c) not a nucleophile 3) what set of reaction conditions should favor an SN2 reaction on 2-bromo-3-methylbutane a) weak nucleophile in a protic solvent b) weal nucleophile in aprotic solvent c) strong nucleophile in a protic solvent d) strong nucleophile in a aprotic solventarrow_forward
- 7. Choose the best reagent from the list below for carrying out each transformation. Place the letter of the reagent in the blank to the left of the reaction. (see attached screenshot). a. 1. O3 2. Zn, H3O+ b. 1. BH3, THF 2. H2O2, NaOH, H2O c. H2O, H2SO4, heat d. 1. OsO4 2. NaHSO3, H2O e. KMnO4, acid f. 1. Hg(OAc)2, H2O 2. NaBH4arrow_forwardWhy Is that the best possible yield? Wittig Reactionarrow_forwardWith reference to the indicated C–H bonds in the following compound:a.) Rank the C–H bonds in order of increasing bond strength. b.) Draw the radical resulting from cleavage of each C–H bond, andclassify it as 1°, 2°, or 3°. c.) Rank the radicals in order of increasing stability.d.) Rank the C–H bonds in order of increasing ease of H abstraction in aradical halogenation reaction.arrow_forward
- Resveratrol is an antioxidant found in the skin of red grapes. Its anticancer, anti-inflammatory, and various cardiovascular effects are under active investigation. (a) Draw all resonance structures for the radical that results from homolysis of the OH bond shown in red. (b) Explain why homolysis of this OH bond is preferred to homolysis of either OH bond in the other benzene ring.arrow_forward6. Identify the best reagent(s) for this reaction. (see attached screenshot). a. H2SO4, HgSO4, H2O b. 1. Disiamylborane, 2. HO–, H2O2 c. K2Cr2O7, H+ d. NaOCl e. H2, Pdarrow_forwardI need help with reaction 2.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

