
(a)
Interpretation:
The molecular geometry, polarity and the strongest intermolecular force present in
Concept introduction:
Geometry of a molecule is the arrangement of atoms of a molecule in space. Geometry is dependent on the number of bond pairs, lone pairs and valence electrons of central atom. Polarity is defined as the tendency of atoms of a molecule to attract the electrons of the bond pairs towards itself.
Answer to Problem 15.3TC
The geometry, polarity and intermolecular force present in
Geometry is tetrahedral.
Polarity of methane is zero. It means
The strongest intermolecular force present is induced dipole.
Explanation of Solution
Methane,
Structure of methane is shown below.
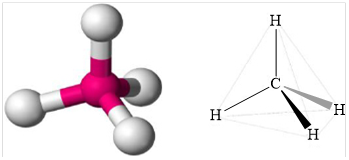
Figure 1
Therefore, methane is tetrahedral, non-polar and strongest intermolecular forces are induced dipole.
Geometry of methane is tetrahedral methane is non polar and the strongest intermolecular force present in methane is induced dipole.
(b)
Interpretation:
The geometry, polarity and the strongest intermolecular force present in
Concept introduction:
Geometry of a molecule of a molecule is the arrangements of atoms of a molecule in space. Geometry is depends on the number of bond pairs, lone pairs and valence electrons of central atom. Polarity is defined as the tendency of atoms of a molecule to attract the bond pairs towards itself.
Answer to Problem 15.3TC
The geometry, polarity and the strongest intermolecular force present in
Geometry is linear.
Polarity of carbon dioxide is zero. It means
Strongest intermolecular force present is induced dipole.
Explanation of Solution
Carbon dioxide
Structure of carbon dioxie is shown below.
![]()
Figure 2
Therefore, carbon dioxide is linear, non-polar and the strongest intermolecular force present is induced dipole.
Geometry of carbon dioxide is linear, polarity is zero means non polar molecule and the strongest intermolecular force present is induced dipole.
(c)
Interpretation:
The geometry, polarity and the strongest intermolecular force present in
Concept introduction:
Geometry of a molecule of a molecule is the arrangements of atoms of a molecule in space. Geometry is depends on the number of bond pairs, lone pairs and valence electrons of central atom. Polarity is defined as the tendency of atoms of a molecule to attract the bond pairs towards itself.
Answer to Problem 15.3TC
The geometry, polarity and intermolecular force present in
Geometry is bent.
Polarity of
Strongest intermolecular force present in
Explanation of Solution
Oxygen difluoride
Structure of oxygen difluoride is shown below.
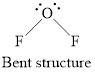
Figure 3
Therefore, oxygen difluoride is bent, polar, and the strongest intermolecular force present is dipole-dipole.
Geometry of oxygen difluoride is bent, polarity is non-zero means polar molecule and the strongest intermolecular force present is dipole dipole interaction.
(d)
Interpretation:
The geometry, polarity and the strongest intermolecular force present in
Concept introduction:
Geometry of a molecule of a molecule is the arrangements of atoms of a molecule in space. Geometry is depends on the number of bond pairs, lone pairs and valence electrons of central atom. Polarity is defined as the tendency of atoms of a molecule to attract the bond pairs towards itself.
Answer to Problem 15.3TC
The geometry, polarity and intermolecular force present in
Geometry is bent.
Polarity of
The strongest intermolecular force present is hydrogen bonding.
Explanation of Solution
Chloric acid
Structure of chloric acid is shown below.
![]()
Figure 4
Therefore, chloric acid is bent, polar and the strongest force present is hydrogen bonding.
Geometry of chloric acid is bent, polarity is non-zero means polar molecule and the strongest intermolecular force present is hydrogen bonding.
Want to see more full solutions like this?
Chapter 15 Solutions
Introductory Chemistry: An Active Learning Approach
- The compounds ethanol (C2H5OH) and dimethyl ether (CH3OCH3) have the same molecular formula. Which is expected to have the higher surface tension? Why?arrow_forwardFor liquid-state samples of the following diatomic molecules, indicate the type or types of intermolecular forces (dipoledipole interactions, hydrogen bonding, London forces) present. There may be more than one correct answer in a given situation. a. H2 b. HF c. CO d. F2arrow_forwardIn which of the following processes is it necessary to break covalent bonds as opposed to simply overcoming intermolecular forces? (a) Decomposing HCI to H2 and Cl2 (b) Dissolving NaCl in water (c) Freezing ethyl alcohol (d) Subliming iodinearrow_forward
- Define the following terms and describe how each depends on the strength of the intermolecular forces. a. surface tension b. viscosity c. melting point d. boiling point e. vapor pressurearrow_forwardWhich of the following compounds would be expected to form intermolecular hydrogen bonds in the liquid state? (a) H2Se (b) HCO2H (formic acid) (c) HI (d) acetone, (CH3)2COarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





