
Interpretation:
How many functional groups present in the Diynemicin A, and all functional groups should be identified given the molecule.
Concept Introduction:
Functional group in
| Group name | Structure | Compound name | Property |
| Hydroxyl | Alcohol | Polar | |
| Carbonyl |
( | Polar | |
| Carboxyl | Acid | ||
| Amino | Polar |
Carbonyl group:
A carbon atom is double-bonded to an oxygen atom
If the carbonyl carbon is attached with two alkyl or aryl group is called as ketone, if the carbonyl carbon is attached with one hydrogen atom and one alkyl or aryl group is called as aldehyde.

Carboxylic acid: One
The derivatives of carboxylic acid are ester, amide
Ester: One
Amide: One
Alcohol:
The compound contains hydroxyl
Example is given below
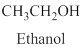
Alkenes:
Alkenes are a class of hydrocarbons. The carbon-carbon double bond is called as alkenes and it is also called as olefins.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
FUNDAMENTALS OF GENERAL CHEM VOL 2
- Draw the four stereoisomers of threonine as Fischer projections.arrow_forwardThreonine has two chiral centers. Draw L-threonine and indicate which carbon atoms are chiral. Which carbon atom is responsible for d and L configuration?arrow_forwardCellulose is water insoluble, despite its many OH groups. Based on its three-dimensional structure, why do you think this is so?arrow_forward
- Trehalose, a disaccharide found in the blood of insects, has the following structure. What simple sugars would you obtain on hydrolysis of trehalose?arrow_forwardMethionine has a sulfur atom in its formula. Explain why methionine does not form disulfide bridges.arrow_forwardDraw the complete structural formula of arachidonic acid (Table 23.1) in a way that shows the cis stereochemistry of its four double bonds.arrow_forward
- One of these forms of cocaine is relatively insoluble inwater: which form, the free base or the hydrochloride?arrow_forwardWhat product is formed when a solution of A and B is treated with mild base? This reaction is the first step in the synthesis of rosuvastatin (sold as a calcium salt under the trade name Crestor), a drug used to treat patients with high cholesterol.arrow_forwardWhich of the two amines, decylamine or ethanamine, would you expect to be more soluble in water and why?arrow_forward
- Why is a thioester bond a “high-energy” bond?arrow_forwardA pentapeptide has a titration curve that shows five inflection points for five equivalents of hydroxide added. How many residues have ionizable groups within their side chains (R-groups) based on this information?arrow_forwardWith respect to the parent compound, explain in detail how modifying the rings shown as A, B, C, D and E in morphine can affect the biological activity associated with the moleculearrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





