
Concept explainers
Interpretation:
For each compound from Table 15-1 that contains only carbon and hydrogen, the number of conjugated
Concept introduction:
A compound containing at least one
Answer to Problem 15.7YT
The value of longest-wavelength
Explanation of Solution
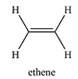
The compounds from Table 15-1 that contain only C and H, the number of conjugated double bonds in them and their longest-wavelength
| Compound | Conjugated |
Compound | Conjugated |
||
| 0 | < 150 | 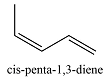 |
2 | 223 | |
 |
0 | 161 | 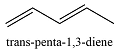 |
2 | 223.5 |
 |
0 | 177 | 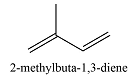 |
2 | 224 |
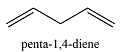 |
0 | 178 | 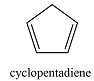 |
2 | 239 |
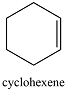 |
0 | 182 | 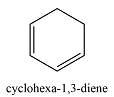 |
2 | 256 |
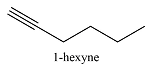 |
0 | 185 | 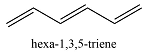 |
3 | 274 |
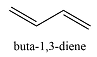 |
2 | 217 | 4 | 290 | |
 |
11 | 455 | |||
For compounds that contain only one double/triple bond – ethene,
As the number of conjugated
The value of the longest-wavelength
Want to see more full solutions like this?
Chapter 15 Solutions
ORG. CHEM. LL W/SSM+SG+ACCESS
- According to the conventions above, what is the sign ( + or ) of the P.E. change (H) for Rxn 3?arrow_forwardPlease help me understand where the 2.54cm comes from to get 7.62•10^-5? This is a lecture problemarrow_forwardIn each of the following molecules 1-8, identify the relation between the circled hydrogen atoms as homotopic, enantiotopic, diastereotopic, heterotopic?arrow_forward
- Define 1,2-alkyl shift ?arrow_forwardFrom the spectra A-J and in the NMR Spectra tile, select the letter that corresponds to 1. methyl butanoate2. benzaldehyde3. 1-chlorobutane4. 1-chloro-2-methylpropane5. butan-2-one6. propan-2-ol7. propanalarrow_forwardwhy are my answers wrong for number 7 help me asap plsarrow_forward
- 4. Why does a carbon to oxygen double bond absorption band have a greater intensity than a carbon to carbon double bond absorption band?arrow_forwardHow do I find the [FeSCN]^2+ value?arrow_forwardCalculate delt G for a.) CH3NH2(aq) + H2O (l) -> CH3NH3+ (aq) + OH-(aq) T= 25C kp= 4.4x 10-4 b.) Pbl2(s) + Pb2+(aq) +2I-(aq) T= 25C Kp= 8.7x10-9arrow_forward
- The discussion this week explores the reasons why the resonance frequency for carbonyl groups covers a large range from 1600 - 1820 cm-1 (learning objective 3). Supply three factors that influence the carbonyl stretching frequency. Discuss in which way these factors contribute to the shift.arrow_forwardHow is this answer found?arrow_forwardShow that a 1,2-methyl shift involving (CH3)3CC+HCH3 is allowed.arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

