
Concept explainers
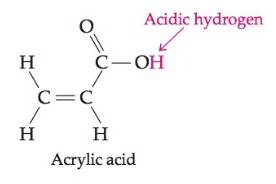
Acrylic acid (
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Chemistry, Books a la Carte Edition and Modified Mastering Chemistry with Pearson eText & ValuePack Access Card (7th Edition)
- Lactic acid, C3H6O3, occurs in sour milk as a result of the metabolism of certain bacteria. Calculate the pH of a solution of 56. mg lactic acid in 250. mL water. Ka for D-lactic acid is 1.5 × 10−4.arrow_forwardBarbituric acid, HC4H3N2O3, is used to prepare barbiturates, a class of drugs used as sedatives. A 325-mL aqueous solution of barbituric acid has a pH of 2.34 and contains 9.00 g of the acid. What is Ka for barbituric acid?arrow_forwardFind the value of Kb for the conjugate base of the following organic acids. (a) picric acid used in the manufacture of explosives; K a =0.16 (b) trichloroacetic acid used in the treatment of warts; K a =0.20arrow_forward
- Consider pyridine, C5H5N, a pesticide and deer repellent. Its conjugate acid, C5H5NH+, has K a =6.7106. (a) Write a balanced net ionic equation for the reaction that shows the basicity of aqueous solutions of pyridine. (b) Calculate Kb for the reaction in (a). (c) Find the pH of a solution prepared by mixing 2.74 g of pyridine in enough water to make 685 mL of solution.arrow_forwardWhat is the pH of a solution obtained by mixing 235 mL of NaOH with a pH of 11.57 and 316 mL of Sr(OH)2 with a pH of 12.09? Assume that volumes are additive.arrow_forwardNicotinic acid, C6H5NO2, is found in minute amounts in all living cells, hut appreciable amounts occur in liver, yeast, milk, adrenal glands, white meat, and corn. Whole wheat (lour contains about 60. 0g per gram of flour. One gram (1.00 g) of the acid dissolves in water to give 60. mL of solution having a pH of 2.70. What is the approximate value of Ka for the acid? Nicotinic acidarrow_forward
- Tartaric acid is a weak diprotic fruit acid with Ka1 = 1.0 103 and Ka2 = 4.6 105. a Letting the symbol H2A represent tartaric acid, write the chemical equations that represent Ka1 and Ka2. Write the chemical equation that represents Ka1 Ka2. b Qualitatively describe the relative concentrations of H2A, HA, A2, and H3O+ in a solution that is about 0.5 M in tartaric acid. c Calculate the pH of a 0 0250 M tartaric acid solution and the equilibrium concentration of [H2A]. d What is the A2 concentration in solutions b and c?arrow_forwardA solution of baking soda, NaHCO3, has a pH of 10.08. What is the percent (by mass) of NaHCO3 in a 235-mL solution? (Assume a density of 1.00 g/mL.)arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





