
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
4th Edition
ISBN: 9781265982959
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 15, Problem 36P
Interpretation Introduction
Interpretation:
Chirality center of dobutamine molecule should be identified and the enantiomers should be drawn.
Concept Introduction:
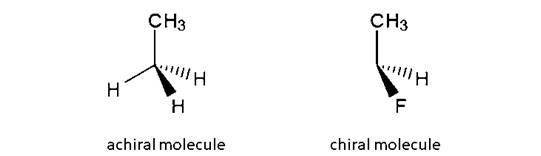
A tetrahedral carbon atom bonded to four different groups is called a chiral center. A Molecule having at least one chiral center is a chiral molecule. Molecules that do not have any chiral centers are called achiral.
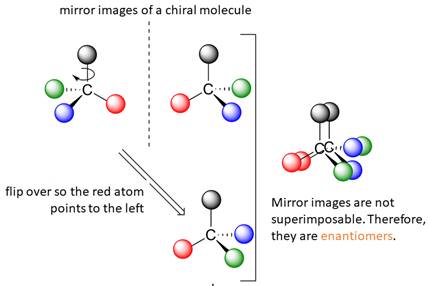
When the mirror images of a chiral molecule are not superimposable, those mirror images become stereoisomers called enantiomers.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Diltiazem is prescribed to treat hypertension, and simvastatin is a cholesterol-lowering drug. Locate the chirality centers in each.
(a) assign R or S configuration to each chiral center,
(b) Which compound are enantiomers?
(c) Which compounds are diastereomers?
Butaclamol is a potent antipsychotic that has been used clinically in the treatment of schizophrenia. How many asymmetric centers does it have?
Chapter 15 Solutions
GENERAL,ORGANIC, & BIOLOGICAL CHEM-ACCES
Ch. 15.1 - Prob. 15.1PCh. 15.1 - Prob. 15.1PPCh. 15.1 - For trans-2-hexene: (a) draw a stereoisomer; (b)...Ch. 15.2 - Prob. 15.3PCh. 15.2 - Prob. 15.4PCh. 15.3 - Prob. 15.2PPCh. 15.3 - Prob. 15.5PCh. 15.3 - Prob. 15.6PCh. 15.3 - Prob. 15.3PPCh. 15.3 - Prob. 15.7P
Ch. 15.3 - Prob. 15.8PCh. 15.3 - Prob. 15.9PCh. 15.4 - Prob. 15.4PPCh. 15.4 - Prob. 15.10PCh. 15.4 - Prob. 15.11PCh. 15.4 - Prob. 15.12PCh. 15.5 - Prob. 15.13PCh. 15.6 - Prob. 15.5PPCh. 15.6 - Prob. 15.14PCh. 15.6 - Prob. 15.15PCh. 15.7 - Prob. 15.16PCh. 15.7 - Prob. 15.17PCh. 15.7 - Prob. 15.6PPCh. 15.7 - Prob. 15.18PCh. 15.8 - Prob. 15.7PPCh. 15.8 - Prob. 15.19PCh. 15.9 - Prob. 15.20PCh. 15 - Prob. 21PCh. 15 - Prob. 22PCh. 15 - Prob. 23PCh. 15 - Prob. 24PCh. 15 - Prob. 25PCh. 15 - Prob. 26PCh. 15 - Prob. 27PCh. 15 - Prob. 28PCh. 15 - Prob. 29PCh. 15 - Prob. 30PCh. 15 - Prob. 31PCh. 15 - Prob. 32PCh. 15 - Prob. 33PCh. 15 - Prob. 34PCh. 15 - Prob. 35PCh. 15 - Prob. 36PCh. 15 - How are the compounds in each pair related? Are...Ch. 15 - Prob. 38PCh. 15 - Prob. 39PCh. 15 - Prob. 40PCh. 15 - Prob. 41PCh. 15 - Prob. 42PCh. 15 - Prob. 43PCh. 15 - Prob. 44PCh. 15 - Prob. 45PCh. 15 - Prob. 46PCh. 15 - Prob. 47PCh. 15 - Prob. 48PCh. 15 - Prob. 49PCh. 15 - Prob. 50PCh. 15 - (a) Define the terms “optically active” and...Ch. 15 - Prob. 52PCh. 15 - Prob. 53PCh. 15 - Prob. 54PCh. 15 - Prob. 55PCh. 15 - Prob. 56PCh. 15 - Prob. 57PCh. 15 - Prob. 58PCh. 15 - Prob. 59PCh. 15 - Prob. 60PCh. 15 - Prob. 61PCh. 15 - Prob. 62PCh. 15 - Prob. 63PCh. 15 - Prob. 64PCh. 15 - Prob. 65PCh. 15 - Prob. 66PCh. 15 - Prob. 67CPCh. 15 - Prob. 68CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Although fresh human sweat has no odor, enzymes in skin bacteria form a variety of compounds with distinctive, pungent odors. The two enantiomers of 3-methyl-3-sulfanyl-1-hexanol (A), which smell somewhat differently, contribute to the characteristic foul odor of sweat. Locate the chirality center in A and draw both enantiomersarrow_forwardLabel the chirality center (if one exists) in each compound. A compound may contain zero or one chirality center.arrow_forwardHow many carbon chirality centers does the molecule possess?arrow_forward
- How many chirality centers are in each compoundarrow_forwardMevacor is used clinically to lower serum cholesterol levels. How many asymmetric centers does Mevacor have?arrow_forwardBystolic (shown below) is a pharmaceutical used to treat high blood pressure. How many asymmetric centers are present in this structure?arrow_forward
- Identify the chirality center (sometimes called chiral atom) in each molecule. If the molecule does not contain a chirality center, select none.arrow_forwardCoibacin B is a natural product that exhibits potent anti-inflammatory activity and potential activity in the treatment of leishmaniasis, a disease caused by certain parasites. Assign the configuration of each chiral center in coibacin B H.arrow_forwardThe quantitative differences in biological activity between the two enantiomers of a compound are sometimes quite large. For example, the D isomer of the drug isoproterenol, used to treat mild asthma, is 50 to 80times more effective as a bronchodilator than the L isomer. Identify the chiral center in isoproterenol. Why do the two enantiomers have such radically different bioactivity?arrow_forward
- Assign an R or S configuration to the chiral center in each molecule.arrow_forwardSimvastatin (trade name: ZocorTM) is a pharmaceutical used to treat elevated blood lipids (i.e. high cholesterol). It contains 7 chirality centers (which allows for 128 different stereoisomers!) Determine the configuration of each chirality center. H H HO H H H H H H H H H O H Harrow_forwardSimvastatin (trade name: Zocor™M) is a pharmaceutical used to treat elevated blood lipids (i.e. high cholesterol). It contains 7 chirality centers (which allows for 128 different stereoisomers!) Determine the configuration of each chirality center. HOL. O CH3 H O H Ou yot H O XII H по / CH3arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning