
Concept explainers
(a)
Interpretation:
Whether the given compound is optically active or not needs to be determined.
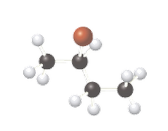
Concept Introduction:
The ball-stick model is a method of representation of molecules to demonstrate the three-dimensional position of atoms and the bonds. The atoms are represented by different colored spheres, while bonds are represented by rods connecting the spheres. Chiral or optically active molecules can rotate plane-polarized light either clockwise or counterclockwise. If plane-polarized light is rotated clockwise then it is known as dextrorotatory and for counterclockwise rotation, it is known as levorotatory.
(b)
Interpretation:
Whether the given compound is optically active or not needs to be determined.
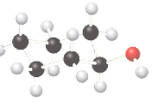
Concept Introduction:
The ball-stick model is a method of representation of molecules to demonstrate the three-dimensional position of atoms and the bonds. The atoms are represented by different colored spheres, while bonds are represented by rods connecting the spheres. Chiral or optically active molecules can rotate plane-polarized light either clockwise or counterclockwise. If plane-polarized light is rotated clockwise then it is known as dextrorotatory and for counterclockwise rotation, it is known as levorotatory.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- One of the analgesics has a chiral center. Which compound is it? One of the two enantiomers is far more effective at reducing pain than the other.arrow_forwardDraw the structure for a compound with molecular formula C2H2I2F2 a. that is optically inactive because it does not have an asymmetric center. b. that is optically inactive because it is a meso compound. c. that is optically active.arrow_forwardWhat is the relationship between the two molecules shown below? OH H CH3 ОН H H H H H Н. H. H. -CH3 H H H H H H A) enantiomers B) diastereomers C) conformational isomers D) constitutional isomers E) identicalarrow_forward
- 1. Give the number of stereogenic centers in the pain reliever Met-enkephalin, mark the corresponding stereogenic centers with an asterisk. H₂N. OH HO S I CH3arrow_forwardLocate the stereogenic centers in each drug. Albuterol is a bronchodilator—that is, it widens airways—so it is used to treat asthma. Chloramphenicol isan antibiotic used extensively in developing countries because of its lowcost.arrow_forwardDetermine whether the following compound is optically active or optically inactive: O optically active O optically inactivearrow_forward
- Identify the relationship between the following two compounds. A) identical B) constitutional isomers C) enantiomers D) diastereomersarrow_forwardLocate the stereogenic centers in each compound. A molecule may have one or more stereogenic centers. Gabapentin enacarbil [part (d)] is used to treat seizures and certain types of chronic pain.arrow_forwardIdentify the relationship between the following two compounds. h A) identical B) constitutional isomers C) enantiomers D) diastereomersarrow_forward
- State how each pair of compounds is related. Are they enantiomers, diastereomers, constitutional isomers, or identical?arrow_forward8. Identify the following sets of compounds as enantiomers, diastereomers, or identical. Relationship Between Compounds (enantiomers, diastereomers, or identical)?arrow_forwardWhat is the relationship between the parent structure on the left and the structure on the right? A: Enantiomers B: Diastereomers C: Constitutional (structural) isomers D: Identical moleculesarrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
