
(a)
Interpretation:
The following pair of molecules should be classified as stereoisomers, constitutional isomers, or identical molecules:
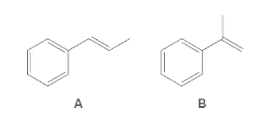
Concept Introduction:
Isomers are the compounds having same molecular formula but different structure. The phenomenon possessed by the molecules is known as isomerism.
Compounds having same molecular formula but possessing different connectivity of atoms or group of atoms are known as constitutional isomers.
The compounds which have same molecular formula, same connectivity of bond but the arrangement of atoms in space is different are known as stereoisomers.
The compounds having same bond connectivity and same molecular formula are known as identical molecules.
(b)
Interpretation:
The following pair of molecule should be classified as stereoisomers, constitutional isomers, or identical molecules:
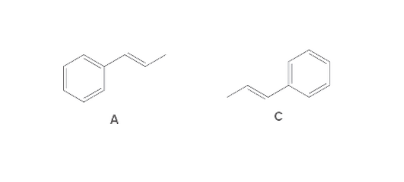
Concept Introduction:
Isomers are the compounds having same molecular formula but different structure. The phenomenon possessed by the molecules is known as isomerism.
Compounds having same molecular formula but possessing different connectivity of atoms are known as constitutional isomers.
The compounds which have same molecular formula, same connectivity of bond but the arrangement of atoms in space is different are known as stereoisomers.
The compounds having same bond connectivity and same molecular formula are known as identical molecules.
(c)
Interpretation:
The following pair of molecule should be classified as stereoisomers, constitutional isomers, or identical molecules:
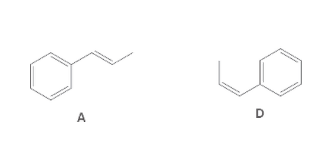
Concept Introduction:
Isomers are the compounds having same molecular formula but different structure. The phenomenon possessed by the molecules is known as isomerism.
Compounds having same molecular formula but possessing different connectivity of atoms are known as constitutional isomers.
The compounds which have same molecular formula, same connectivity of bond but the arrangement of atoms in space is different are known as stereoisomers.
The compounds having same bond connectivity and same molecular formula are known as identical molecules.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- Consider the tricyclic structure A. (a) Label each substituent on the rings as axial or equatorial. (b) Draw a skeletal structure for A, using wedges and dashes to show whether the substituents are located above or below the rings.arrow_forwardChondrocole A is a marine natural product isolated from red seaweed that grows in regions of heavy surf in the Pacific Ocean. (a) Predict the solubility of chondrocole A in water and CH2Cl2. (b) Locate the stereogenic centers and label each as R or S. (c) Draw a stereoisomer and a constitutional isomer of chondrocole A.arrow_forwardNonearrow_forward
- Question a, b & carrow_forwardClassify each pair of compounds as constitutional isomers, stereoisomers, identical molecules, or not isomers of each other.arrow_forwardConsider the tricyclic structure A. (a) Label each substituent on the rings as axial or equatorial. (b) Draw a skeletal structure for A, using wedges and dashed wedges to show whether the substituents are located above or below the rings.arrow_forward
- 22..Draw a constitutional isomer for the following compound. Y Csindo Jenido torarrow_forwardQ2: Drawn are four isomeric dimethylcyclopropanes. a. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and 8; A and C; B and C, Cand D? b. Label each compound as chiral or achiral. c. Which compounds, alone, would be optically active? d. Which compounds have a plane of symmetry? e. Which of the compounds are meso compounds? f. Would an equal mixture of compounds C and D be optically active? What about an equal mixture of B and C?arrow_forwardHow are the ball-and-stick models A and B related to each other? Do they represent constitutional isomers or identical molecules?arrow_forward
- 6. What is the relationship between A and B? HO OH II A) They are constitutional isomers C) They are tautomers B) D) They are stereoisomers They are enantiomersarrow_forward0 Draw the fi ve constitutional isomers having molecular formula C 6H 14.arrow_forwardConsider the ball-and-stick models A–D. How is each pair of compounds related: (a) A and B; (b) A and C; (c) A and D; (d) C and D? Choose from identical molecules, enantiomers, or diastereomers.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning




