
Concept explainers
(a)
Interpretation:
The following compound should be labeled as optically active or inactive:
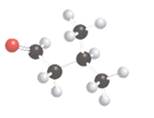
Concept Introduction:
For an optically inactive compound, there should be no chiral carbon present in the molecule that is the carbon should be bonded to two or more same atom or group of atoms. For an optically active compound, there should be a chiral carbon present in the molecule that is the carbon should be bonded to four different atom or group of atoms.
(b)
Interpretation:
The following compound should be labeled as optically active or inactive:
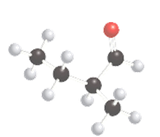
Concept Introduction:
For an optically inactive compound, there should be no chiral carbon present in the molecule that is the carbon should be bonded to two or more same atom or group of atoms. For an optically active compound, there should be a chiral carbon present in the molecule that is the carbon should be bonded to four different atom or group of atoms.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- (a) Locate the stereogenic centers in the ball-and-stick model of ezetimibe (trade name Zetia), a cholesterol-lowering drug. (b) Label each stereogenic center as R or S. ezetimibearrow_forwardLabel each of the following structures as a cyclic hemiacetal, hemiketal, acetal, ketal, or none of these: a. b. c.arrow_forwardClassify each compound as IDENTICAL to A or an ENANTIOMER to A.arrow_forward
- Draw the structure of A,B,and Carrow_forwardGive the IUPAC name for each compound, including the R, S designation for each stereogenic center. In a. b. C.arrow_forwardHow are the compounds in each pair related to each other? Are they identical, enantiomers, diastereomers, constitutional isomers, or not isomers of each other? a. b. H НО H 'OH H and and H H H HO OHarrow_forward
- Captopril is a drug used to treat high blood pressure and congestiveheart failure.Designate each stereogenic center as R or S.arrow_forwardInstructions: Label each pair of stereoisomers as:a. enantiomersb. diastereomersc. identicalarrow_forwardProblem 5.16 Label each stereogenic center as R or S. e. HO, HO H f.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

