
a)
Interpretation:
Energy consumed by an average human per day, which is 2100Kcal,should be converted intowatts.
Concept Introduction:
As per the data given, the inventors tried to compare the output of steam engines as that of the horses.The engine would replace the horses. An average workhorse expends energy at a rate of which is originally defined as 550-foot pounds/second and it means, it could pull 550pounds through a distance in one foot in one sec., whereas in modern units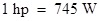 Therefore, a modern race car with 2.4-L V8 engine is about 740hp.
Therefore, a modern race car with 2.4-L V8 engine is about 740hp.
b)
Interpretation:
Ratio of horsepower to human power should be derived.
Concept Introduction:
As per the data given, the inventors tried to compare the output of steam engines as that of the horses. The engine would replace the horses. An average workhorse expends energy at a rate of which is originally defined as 550-foot pounds/second and it means, it could pull 550 pounds through a distance in one foot in one sec., whereas in modern units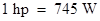 Therefore, a modern race car with 2.4-L V8 engine is about 740 hp.
Therefore, a modern race car with 2.4-L V8 engine is about 740 hp.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Chemistry For Changing Times (14th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





