
Interpretation:
To give the explanation whether the acid-catalyzed hydrolysis of acetamide is a reversible or irreversible reaction.
Concept introduction:
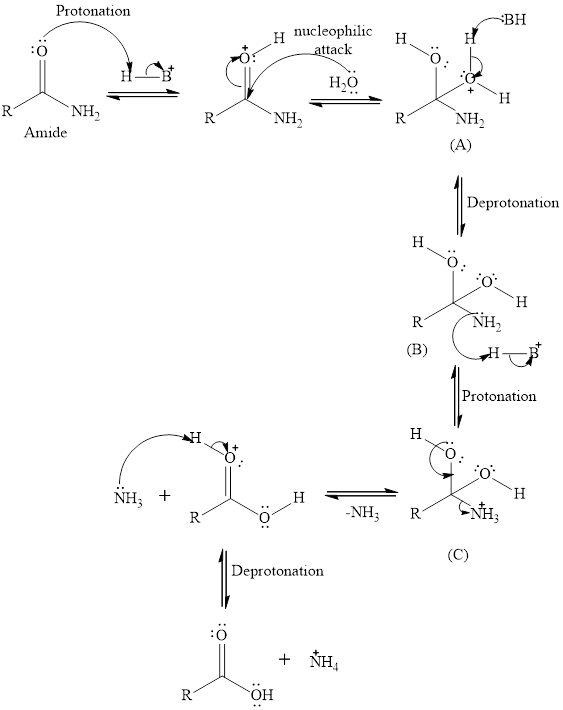
In acid hydrolysis of amide, acid protonates carbonyl oxygen of amide which increases the electrophilicity of the carbonyl carbon. Water acts as a nucleophile gets attached to the carbonyl carbon of amide to form tetrahedral intermediate(A) which on losing proton get converted to tetrahedral intermediate(B). The nitrogen of tetrahedral intermediate (B) gets protonated to form tetrahedral intermediate(C). The
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry; Organic Chemistry Study Guide A Format: Kit/package/shrinkwrap
- What is the Methods used to Synthesis of IMINES.arrow_forward1.Explain what is anomeric effect and why it happens? 2.Explain how a glycosyl acetimidate can loss its glycosyl donor activity?arrow_forwardKetoprofen and ibuprofen both have a propanoic acid substituent. Explain why the identical subunits are synthesized in different ways.arrow_forward
- Which of the following substituent is an ortho/para-directing activator and an ortho/para-directing deactivator? a amino group b halogen group c nitro group d carboxyl grouparrow_forward6. What are the most likely impurities present in synthesised metacetamolarrow_forwardWhich of the following substituent is a meta-directing deactivator? a amino group b halogenation group c phenyl group d carboxyl grouparrow_forward
- Indicate whether the following statement is true or false . Benzenesulfonyl chloride or p-toluenesulfonylchloride give N-substituted sulfonamides with primary and secondary amines. The derivative consisting of primary amines is insoluble in dilute NaOH.RightFalsearrow_forwardThe pathway for nicotinic acid requires a series of highly unfavorable reactions. Which other alkaloid molecular scaffold would you use to arrive to such a molecule? And, which series of hypothetical enzymes would you use?arrow_forwardAssume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material and that the mevalonate pathway is followed. Identify the positions in -cadinol where the label would appear.arrow_forward
- A strong acid is generally used to catalyze the Fischer esterification of carboxylic acids.What two steps in the reaction are accelerated by the presence of strong acid? What function does the acid play in each of these steps?arrow_forwardWrite a structural formula for the imine formed in each reactionarrow_forward

 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


