
Concept explainers
Interpretation:
To predict the structure of the coloured compound when the given colourless compound come in contact with the acid treated sheet.
Concept introduction:
Acid-base indicators are the compounds which changetheir colour on changing the pH. The reaction of the acid-base indicator can be represented by:
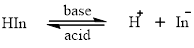
In organic compounds chromophoreis the unsaturated part that imparts colour. An auxochrome is a functional group attached to the chromophore which enhances its property. The increased pi-conjugation decreases the energy gap between pi bonding and pi anti-bonding orbital. The energy absorbs by the electron to get excited from pi bonding to pi anti-bonding orbital get decreased. The wavelength of the emitted photon is in the visible region so the compound is intensely coloured.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic Chemistry; Organic Chemistry Study Guide A Format: Kit/package/shrinkwrap
- Suppose you have a mixture of these three compounds. Devise a chemical procedure based on their relative acidity or basicity to separate and isolate each in pure form.arrow_forwardPredict the products obtained from the reaction of triolein with the following reagents.(a) NaOH in water (b) H2 and a nickel catalyst (c) Br2 in CCl4arrow_forwardWhich of the following is not one of the features of EDTA?A. It is an aminocarboxylic acid with 6 functional groups.B. Soluble in water and polar solvents.c. Its most important feature is that it can form water-soluble complexes with polyvalent metal ions over a wide range of pH.d.The most important feature of D. EDTA is its low capacity to make complex.arrow_forward
- Which of the following is not one of the features of EDTA? A. It is an aminocarboxylic acid with 6 functional groups. B. Soluble in water and polar solvents. C. Its most important feature is that it can form water-soluble complexes with polyvalent metal ions over a wide range of pH. The most important feature of D. EDTA is its low capacity to make complex.arrow_forwardWhich of the following is not one of the features of EDTA? A. It is an aminocarboxylic acid with 6 functional groups. B. Soluble in water and polar solvents. C-Its most important feature is that it can form water-soluble complexes in a wide range of pH with polyvalent metal ions. d. The most important feature of EDTA is its low capacity to make complex.arrow_forwardTreating chitin with H2O, -OH hydrolyzes its amide linkages, forming a compound called chitosan. What is the structure of chitosan? Chitosan has been used in shampoos, bers for sutures, and wound dressings.arrow_forward
- Predict the products obtained from the reaction of triolein with the following reagents.(a) NaOH in water (b) H2 and a nickel catalystarrow_forwardWhat happens when the codeine molecule (C18H21NO3) is exposed to air? What reactions are obtained?arrow_forwardd-Glucuronic acid is found widely in plants and animals. One of its functions is to detoxify poisonous HO-containing compoundsby reacting with them in the liver to form glucuronides. Glucuronides are water soluble and therefore readily excreted. Afteringestion of a poison such as turpentine or phenol, the glucuronides of these compounds are found in the urine. Draw thestructure of the a- and b-glucuronides formed by the reaction of b-d-glucuronic acid and phenol.arrow_forward
- Stearic acid: Description of bromine (red-orange) color persistancearrow_forwardCholic acid is secreted in bile as an amide linked to the aminogroup of glycine. This cholic acid–amino acid combination acts asan emulsifying agent to disperse lipids in the intestines for easierdigestion. Draw the structure of the cholic acid–glycine combination,and explain why it is a good emulsifying agent.arrow_forwardDigitalis is a preparation made from the dried seeds and leaves of the purple foxglove, Digitalis purpurea, a plant native to southern and central Europe and cultivated in the United States. The preparation is a mixture of several active components, including digitalin. Digitalis is used in medicine to increase the force of myocardial contraction and as a conduction depressant to decrease heart rate (the heart pumps more forcefully but less often).arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



