
Figure Q15.8 shows a P-versus-V graph for two processes, A and B, that are performed with the same amount of ideal gas Process A is an isothermal process. Which of the following statements are correct? (More than one statement may be correct.)
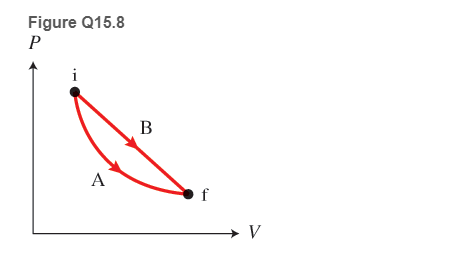
a. The final and the initial temperatures in process B can be different than in process A.
b. The final and the initial temperatures in process B are always the same as in process A.
c. The work done by the gas on the environment is the same in both processes.
d. The work done by the gas on the environment in process B is greater than that in process A.
e. The work done by the gas on the environment in process B is smaller than that in process A.
f. The temperature of the gas in process B is first increasing and then decreasing.
g. The temperature of the gas in process B is first decreasing and then increasing.
h. The temperature of the gas in process B is constant.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
College Physics
Additional Science Textbook Solutions
Tutorials in Introductory Physics
Conceptual Physics (12th Edition)
An Introduction to Thermal Physics
College Physics (10th Edition)
Conceptual Physical Science (6th Edition)
Life in the Universe (4th Edition)
- (a) If you shake a jar full of jelly beans of different sizes, the larger beans tend to appear near the top and the smaller ones tend to fall to the bottom. Why? (b) Does this process violate the second law of thermodynamics?arrow_forwardIn a cylinder of an automobile engine, immediately after combustion the gas is confined to a volume of 50.0 cm3 and has an initial pressure of 3.00 106 Pa. The piston moves outward to a final volume of 300 cm3, and the gas expands without energy transfer by heat, (a) What is the final pressure of the gas? (b) How much work is done by the gas in expanding?arrow_forwardA sample of an ideal gas goes through the process shown in Figure P17.26. From A to B, the process is adiabatic; from B to C, it is isobaric with 100 kJ of energy entering the system by heat; from C to D, the process is isothermal; and from D to A, it is isobaric with 150 kJ of energy leaving the system by heat. Determine the difference in internal energy Eint,B Eint,A.arrow_forward
- (a) Calculate the rate of heat transfer by radiation from a car radiator at 110C into a 50.0C environment, if the radiator has an emissivity of 0.750 and a 1.20m2 surface area. (b) Is this a significant fraction of the heat transfer by an automobile engine? To answer this, assume a horsepower of 200 hp (1.5 kW) and the efficiency of automobile engines as 25%.arrow_forwardPractical steam engines utilize 450C steam, which is later exhausted at 270C. (a) What is the maximum eficiency that such a heat engine can have? (b) Since 270C steam is still quite hot, at second steam engine is sometimes operated using the exhaust of the first. What is the maximum eficiency of the second engine if its exhaust has a temperature of 150C ? (c) What is the overall eficiency of the two engines? (d) Show that this is the same eficiency as a single Carnot engine operating between 450C and 150C. Explicitly show how you follow the steps in the Problem-Solving Strategies for Thermodynamics.arrow_forward(a) In reaching equilibrium, how much heat transfer occurs from 1.00 kg of water at 40.0C when it is placed in contact with 1.00 kg of 20.0C water in reaching equilibrium? (b) What is the change in entropy due to this heat transfer? (c) How much work is made unavailable, taking the lowest temperature to be 20.0C ? Explicitly show how you follow the steps in the Problem-Solving Strategies for Entropy.arrow_forward
- Unreasonable Results (a) Suppose you want to design a steam engine that has heat transfer to the environment at 270C and has a Carnot eficiency of 0.800. What temperature of hot Steam must you use? (b) What is unreasonable about the temperature? (c) Which premise is unreasonable?arrow_forwardUnreasonable Results (a) What is the temperature increase of an 80.0 kg person who consumes 2500 kcal of food in one day with 95.0% of the energy transferred as heat to the body? (b) What is unreasonable about this result? (c) Which premise or assumption is responsible?arrow_forwardOne mole of an ideal gas is contained in a cylinder with a movable piston. The initial pressure, volume, and temperature are Pi, Vi, and Ti, respectively. Find the work done on the gas in the following processes. In operational terms, describe how to carry out each process and show each process on a PV diagram. (a) an isobaric compression in which the final volume is one-half the initial volume (b) an isothermal compression in which the final pressure is four times the initial pressure (c) an isovolumetric process in which the final pressure is three times the initial pressurearrow_forward
 Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning





