
Concept explainers
(a)
Interpretation:
To write the mechanism for an uncatalyzed hydrolysis of methyl propionate.
Concept introduction:
The hydrolysis reaction of etser is the reaction in which the breaking of ester bond is done by using water molecule. The product obtained after the ester hydrolysis is the corresponding
The general reaction of an ester to produce in presence of only water is written as,
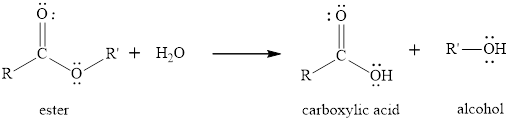
(b)
Interpretation:
To write the mechanism for aminolysis of phenyl formate using methyl amine.
Concept introduction:
The reaction of a carboxylic acid derivative with ammonia, primary and secondary
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
ORGANIC CHEMISTRY-W/S.G+SOLN.MANUAL
- 12. Murexid test as a group-wide reaction to purine derivatives is based on chemical processes: A. Hydrolytic decomposition B. Electrophilic substitution C. Oxidation D. Nucleophilic additionarrow_forwarda. Reaction of acyl compounds with LiAlH4 forms alcohols. b.Reaction of acyl compounds with alcohols forms esters. c.Reaction of carbonyl compounds with LiAlH4 forms alcohols. d.Reaction of carbonyl compounds with alcohols forms hemiacetals. Which are false?arrow_forwardGive the formula and enumerate the functions of the following carboxylic acids: a. Formic acid b. Citric acid c. Butyric acid d. Pimelic acidarrow_forward
- 10. Under certain conditions interacts with sodium nitrite: A. Resorcinol B. Nicotinic acid C. Butadion D. Morphine hydrochloride E. Benzoic acidarrow_forwardWhich could explain the stronger acidity of phenols compared to alcohols. Why? a.pi-electron delocalization b.steric effect c.hydrogen bonding d.hyperconjugationarrow_forwardWhich of the following is the most active acyl compound in terms of susceptibility to react? A. acetic acid B. acetic anhydride C. acetyl chloride D. acetamide E. ethylacetatearrow_forward
- Define the following terms: a. reaction mechanism b. carbocation c. enediol d. general acid e. general basearrow_forward27. What reaction converts the C=O bond to the C=C bond? a. Witten Reaction b. Wittig Reaction c. Winding Reaction d. Whinging Reaction 28. What reagent can reduce imines to primary amines? a. Lithium aluminiumhydride b. Phosphorous pentachloride c. Pyridinium chlorochromate d. Sodium cyanoborohydride 29. What are the steps that can convert imines to amino acids? a. 1. LiAlH₄; Water b. 1. NaCN; 2. Water c. 1. LiAlH₄; 2. Ammonia d. 1. NaCN; 2. Ammonia 30. Cyclohexanone reacts with NaCN to form a.The S-isomer b.The R-isomer c.A racemic mixture d.None of the abovearrow_forwardWhich of the following is the most active acyl compound in terms of susceptibility to react? A. ethylacetateB. acetamideC. acetic anhydrideD. acetyl chlorideE. acetic acidarrow_forward
- 61arrow_forwardWrite the chemical equations involved in the following reactions: a. acetic acid plus ethyl alcohol in the presence of conc. H2SO4 b. acetaldehyde plus Tollens’ reagentarrow_forwardReduction of an alkyl azide results in the formation of —-. A. an imine B. an oxime C. a tertiary amine D. a secondary amine E. a primary aminearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

