
Concept explainers
Catalytic antibodies catalyze a reaction by forcing the conformation of the substrate in the direction of the transition state. The synthesis of the antibody is carried out in the presence of a transition state analog—a stable molecule that structurally resembles the transition state. This causes an antibody to be generated that recognizes and binds to the transition state, thereby stabilizing it. For example, the following transition state analog has been used to generate a catalytic antibody that catalyzes the hydrolysis of the structurally similar ester:
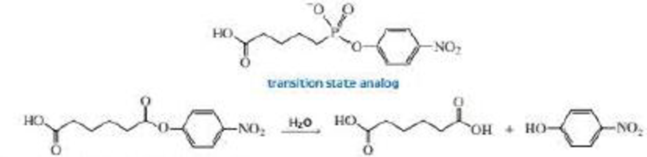
a. Draw a possible transition state for the hydrolysis reaction.
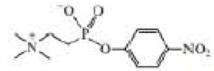
b. The following transition state analog is used to generate a catalytic antibody for the catalysis of ester hydrolysis. Draw the structure of an ester whose rate of hydrolysis would he increased by this catalytic antibody.
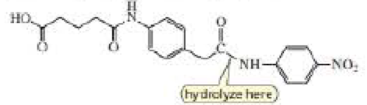
c. Design a transition state analog that catalyzes amide hydrolysis at the amide group indicated.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
ORGANIC CHEMISTRY-W/S.G+SOLN.MANUAL
- What do you predict the addition of a base will have on catalase? Will it increase, decrease, or have no effect on the chemical reaction between hydrogen peroxide and catalase?arrow_forwardExplain how an enzyme is able to catalyze the synthesis of of a large molecule from two smaller molecules.arrow_forwardThe substrate dissociation constant, Ks, is k-1/k1. What do large values of this constant mean? That the formation of product will be fast Strong binding between enzyme and substrate that the reaction is unimolecular Weak binding between enzyme and substratearrow_forward
- List two ways that enzyme catalysis of a reaction is superior to normal conditions.arrow_forwardThe hydrolysis of an ester can be sped up by both acidic and basic conditions. Aminolysis of an ester can be sped up by acidic conditions, but not by basic conditions. Explain why.arrow_forwardThe Michaelis-Menten equation is an expression of the relationship between the initial velocity Vo of an enzymatic reaction and substrate concentration [S]. There are three conditions that are useful for simplifying the Michaelis-Menten equation to an expression from which the effect of [S] on the rate can be more readily determined. Match the condition (e.g., [S] = Km) with the statement or statements that describe it. Doubling [S] will almost double the rate. Half of the active sites are occupied by substrate. About 90% of the active sites are occupied by substrate. Doubling [S] will have little effect on the rate. Less than 10% of the active sites are occupied by substrate. This condition will result in the highest rate. Answer Bank [S] = 0.1 Km [S] = Km [S] = 10 Kmarrow_forward
- Whethor or note the reaction will take place or not depends mainly on? 1. intricate structure of the enzyme. 2. equilibrium of a system. 3. presence of coenzyme. 4. amount of available substrate. 5. Gibbs free energy differences.arrow_forwardOne of the free energy (G)-reaction coordinate graphs given below belong to a ligand-binding protein, while the other two belong to two different enzymes. A)Which of the given graphs cannot belong to an enzyme? A, B or C Choose one of the options and explain your reasoning in one or two sentences. B)What does it mean if an enzyme has reached catalytic perfection? means? Which of the graphs of free energy change given above Could it belong to a catalytically perfect enzyme? C)what kinetic parameter/combination of parameters an enzyme shows its efficiency / whether it has reached catalytic perfection? D)Which step in the Michaelis-Menten mechanism is the rate of diffusion? limited to?arrow_forwardThe following experimental data were collected during a study of the catalytic activity of an intestinalpeptidase with the substrate glycylglycine: Glycylglycine + H2O → 2 glycine [S] (mM) Rate of product formation (mol/min) 1.5 0.21 2.0 0.24 3.0 0.28 4.0 0.33 8.0 0.40 16.0 0.45 Use the graphical analysis (Lineweaver-Burk plot and equation) to determine the Km and Vmax for this enzyme preparation and substrate.arrow_forward
- Let's try similar things with another enzyme! Here's one with a cool mechanism that makes an interesting product. Cys157 Cys157 Cys157 His296 His296 His296 OH HNN: HN, HN -CO2 CH2 Asn329 Asn329 Asn329- Cys157 His296 OH OH HNNH HC. Asn329 N Note that this figure is typical of those found in the biochemical literature in that it provides a somewhat abbreviated view of what is happening. For example, the arrows I have circled in red above for the fırst step need some sorting out. What mechanism is represented by these arrows? O a two step, addition/elimination reaction O an SN2 reaction O an SN1 reaction O nucleophilic aromatic substitution O electrophilic aromatic substitutionarrow_forwardPlease draw the complete, detailed mechanism of the reaction step by step.arrow_forwardOxaloacetate is an inhibitor of succinate dehydrogenase because it is structurally very similar to succinate as shown below. Also, its binding to the enzyme does not involve any covalent bond formation. What type of inhibitor is oxaloacetate? COO | CH₂ 1 CH₂ 1 COO Succinate COO | CH₂ C=O COO Oxaloacetate O a noncompetitive inhibitor both a noncompetitive and an irreversible inhibitor O an irreversible inhibitor O a competitive inhibitorarrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





