
Draw all reasonable resonance structures for each species.
a.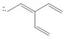 b.
b. c.
c.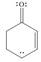 d.
d.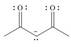 e.
e.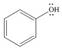 f.
f. 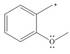
(a)
Interpretation: The possible resonance structure for the given species is to be drawn.
Concept introduction: Most of the organic structures cannot be represented using single Lewis structure. Therefore, there exists more than one Lewis structure for representing a molecule or ion. These structures are known as resonance structures. These are the hypothetical structures and do not specify the exact structure. These resonance structure combine together to give resonance hybrid that is lower in energy and is the most stable structure.
The delocalization of electrons results in the formation resonance structure.
Answer to Problem 16.32P
The possible resonance structure for the given species is,
Explanation of Solution
The method by which overall delocalization of electrons can be described in a particular molecule is known as resonance.
The given species is shown below.
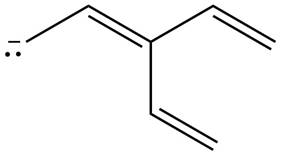
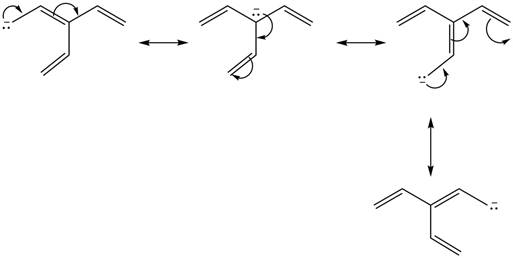
Figure 1
Delocalization of electrons from one position to the other position result in the formation of resonance structure. The resonance structure for the given species is shown below.
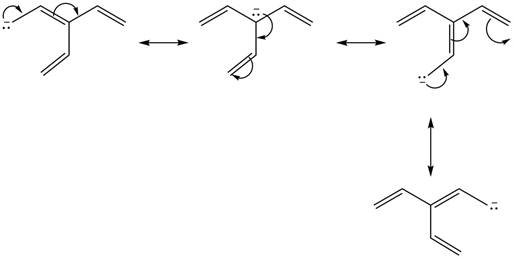
Figure 2
The possible resonance structure for the given species is shown in Figure 2.
(b)
Interpretation: The possible resonance structure for the given species is to be drawn.
Concept introduction: Most of the organic structures cannot be represented using single Lewis structure. Therefore, there exists more than one Lewis structure for representing a molecule or ion. These structures are known as resonance structures. These are the hypothetical structures and do not specify the exact structure. These resonance structure combine together to give resonance hybrid that is lower in energy and is the most stable structure.
The delocalization of electrons results in the formation resonance structure.
Answer to Problem 16.32P
The possible resonance structure for the given species is,
![]()
Explanation of Solution
The method by which overall delocalization of electrons can be described in a particular molecule is known as resonance. Carbocation shows resonance structures when double bond is present in conjugation with it.
The given species is shown below.
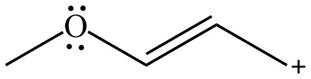
Figure 3
Delocalization of electrons from one position to the other position result in the formation of resonance structure. The resonance structure for the given species is shown below.
![]()
Figure 4
The possible resonance structure for the given species is shown in Figure 4.
(c)
Interpretation: The possible resonance structure for the given species is to be drawn.
Concept introduction: Most of the organic structures cannot be represented using single Lewis structure. Therefore, there exists more than one Lewis structure for representing a molecule or ion. These structures are known as resonance structures. These are the hypothetical structures and do not specify the exact structure. These resonance structure combine together to give resonance hybrid that is lower in energy and is the most stable structure.
The delocalization of electrons results in the formation resonance structure.
Answer to Problem 16.32P
The possible resonance structure for the given species is,
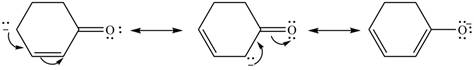
Explanation of Solution
The method by which overall delocalization of electrons can be described in a particular molecule is known as resonance.
The given species is shown below.
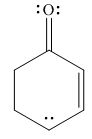
Figure 5
Delocalization of electrons from one position to the other position result in the formation of resonance structure. The resonance structure for the given species is shown below.
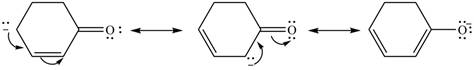
Figure 6
The possible resonance structure for the given species is shown in Figure 6.
(d)
Interpretation: The possible resonance structure for the given species is to be drawn.
Concept introduction: Most of the organic structures cannot be represented using single Lewis structure. Therefore, there exists more than one Lewis structure for representing a molecule or ion. These structures are known as resonance structures. These are the hypothetical structures and do not specify the exact structure. These resonance structure combine together to give resonance hybrid that is lower in energy and is the most stable structure.
The delocalization of electrons results in the formation resonance structure.
Answer to Problem 16.32P
The possible resonance structure for the given species is,
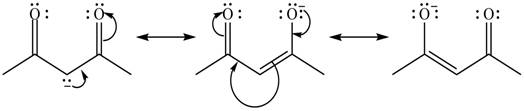
Explanation of Solution
The method by which overall delocalization of electrons can be described in a particular molecule is known as resonance.
The given species is shown below.
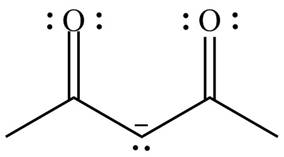
Figure 7
Delocalization of electrons from one position to the other position result in the formation of resonance structure. The resonance structure for the given species is shown below.

Figure 8
The possible resonance structure for the given species is shown in Figure 8.
(e)
Interpretation: The possible resonance structure for the given species is to be drawn.
Concept introduction: Most of the organic structures cannot be represented using single Lewis structure. Therefore, there exists more than one Lewis structure for representing a molecule or ion. These structures are known as resonance structures. These are the hypothetical structures and do not specify the exact structure. These resonance structure combine together to give resonance hybrid that is lower in energy and is the most stable structure.
The delocalization of electrons results in the formation resonance structure.
Answer to Problem 16.32P
The possible resonance structure for the given species is,
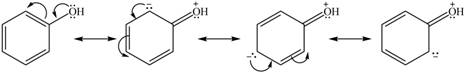
Explanation of Solution
The method by which overall delocalization of electrons can be described in a particular molecule is known as resonance.
The given species is shown below.
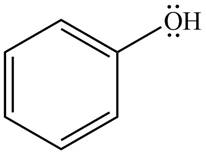
Figure 9
Delocalization of electrons from one position to the other position result in the formation of resonance structure. The resonance structure for the given species is shown below.
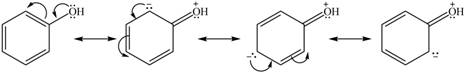
Figure 10
The possible resonance structure for the given species is shown in Figure 10.
(f)
Interpretation: The possible resonance structure for the given species is to be drawn.
Concept introduction: Most of the organic structures cannot be represented using single Lewis structure. Therefore, there exists more than one Lewis structure for representing a molecule or ion. These structures are known as resonance structures. These are the hypothetical structures and do not specify the exact structure. These resonance structure combine together to give resonance hybrid that is lower in energy and is the most stable structure.
The delocalization of electrons results in the formation resonance structure.
Answer to Problem 16.32P
The possible resonance structure for the given species is,
Explanation of Solution
The method by which overall delocalization of electrons can be described in a particular molecule is known as resonance.
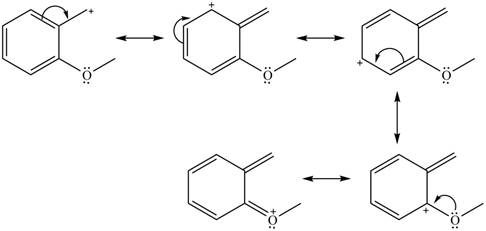
The given species is shown below.
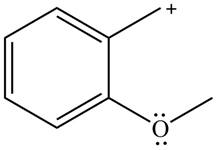
Figure 11
Delocalization of electrons from one position to the other position result in the formation of resonance structure. The resonance structure for the given species is shown below.

Figure 12
The possible resonance structure for the given species is shown in Figure 12.
Want to see more full solutions like this?
Chapter 16 Solutions
ORGANIC CHEMISTRY W/CONNECT PKG
- Question : What are the structure of A, B and C from the image attached.arrow_forwardUse Huckel’s rule to identify if its aromatic or not. Draw the resonance structure of the ff. compounds. note: I need the answer immediately. I will send a good rate right away as well.arrow_forwardDraw ALL REASONABLE resonance structures and the resonance hybrids of the compounds below.arrow_forward
- Tell whether each compound as aromatic, nonaromatic, or antiaromatic. Give explanation.arrow_forwardexplain part A by first drawing all the resonance structures for the four compounds please. and then write the explanaiton. HANDDRAW it instead of using text. thanksarrow_forwardQuestion (Organic Chemistry) Consider molecules A–B. Does the arrow pushing in each structure leadto an acceptable resonance form? If so, explain why it is acceptable or unacceptable? What are the rules and logic? If so, draw it and carefully explain your answer and provide explanations for why each structure leds to an acceptable or unacceptable resonance form. Why is the arrow pushing acceptable or unacceptable in each individual case? Explain you answer with many details, logic, and in a step-by-step fashion.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





