
ORGANIC CHEMISTRY W/CONNECT PKG
5th Edition
ISBN: 9781260901269
Author: SMITH
Publisher: MCG CUSTOM
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 16.46P
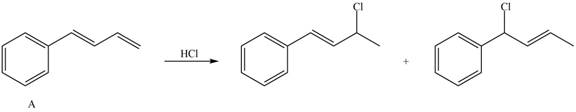
Explain, with reference to the mechanism, why addition of one equivalent of HCl to diene A forms only two products of electrophilic addition, even though four constitutional isomers are possible.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Complete the following by drawing products for a and b, and also identify the diene and dienophile in each one.
Draw and discuss the mechanism (with arrows to show electron movements) of the Diels-Alder reaction between anthracene and maleic anhydride. Draw the orientation and phases of the reacting p-orbitals showing how they overlap in a “suprafacial” geometry to form product
Explain why the ratio of reactants of Nitration of Bromobenzene and reactants of friedel-crafts alkylation of p-Xylene with 1-Bromopropane are different.
Chapter 16 Solutions
ORGANIC CHEMISTRY W/CONNECT PKG
Ch. 16 - Prob. 16.1PCh. 16 - Prob. 16.2PCh. 16 - Problem 16.3 Draw a second resonance structure for...Ch. 16 - Prob. 16.4PCh. 16 - Problem 16.5 Farnesyl diphosphate is synthesized...Ch. 16 - Prob. 16.6PCh. 16 - Prob. 16.7PCh. 16 - Prob. 16.8PCh. 16 - Problem 16.9 Determine the hybridization of the...Ch. 16 - Problem 16.10 Draw the structure consistent with...
Ch. 16 - Problem 16.11 Neuroprotectin D1 (NPD1) is...Ch. 16 - Problem 16.12 Using hybridization, predict how the...Ch. 16 - Problem 16.13 Use resonance theory to explain why...Ch. 16 - Prob. 16.14PCh. 16 - Prob. 16.15PCh. 16 - Problem 16.16 Draw the products formed when each...Ch. 16 - Problem 16.17 Draw a stepwise mechanism for the...Ch. 16 - Prob. 16.18PCh. 16 - Problem 16.19 Draw the product formed when each...Ch. 16 - Prob. 16.20PCh. 16 - Prob. 16.21PCh. 16 - Problem 16.22 Rank the following dienophiles in...Ch. 16 - Prob. 16.23PCh. 16 - Prob. 16.24PCh. 16 - Problem 16.25 What diene and dienophile are needed...Ch. 16 - Prob. 16.26PCh. 16 - Problem 16.27 Which compound in each pair absorbs...Ch. 16 - Prob. 16.28PCh. 16 - 16.29 Name each diene and state whether the...Ch. 16 - Prob. 16.30PCh. 16 - 16.31 Which of the following systems are...Ch. 16 - 16.32 Draw all reasonable resonance structures for...Ch. 16 - Prob. 16.33PCh. 16 - Prob. 16.34PCh. 16 - 16.35 Explain why the cyclopentadienide anion A...Ch. 16 - Prob. 16.36PCh. 16 - 16.37 Draw the structure of each compound.
a. in...Ch. 16 - Prob. 16.38PCh. 16 - 16.39 Label each pair of compounds as...Ch. 16 - Prob. 16.40PCh. 16 - 16.41 Draw the products formed when each compound...Ch. 16 - Prob. 16.42PCh. 16 - 16.43 Treatment of alkenes A and B with gives the...Ch. 16 - 16.44 Draw a stepwise mechanism for the following...Ch. 16 - Prob. 16.45PCh. 16 - 16.46 Explain, with reference to the mechanism,...Ch. 16 - Prob. 16.47PCh. 16 - Prob. 16.48PCh. 16 - Prob. 16.49PCh. 16 - Prob. 16.50PCh. 16 - Prob. 16.51PCh. 16 - Prob. 16.52PCh. 16 - 16.53 Diels–Alder reaction of a monosubstituted...Ch. 16 - Prob. 16.54PCh. 16 - 16.55 Devise a stepwise synthesis of each compound...Ch. 16 - Prob. 16.56PCh. 16 - 16.57 A transannular Diels–Alder reaction is an...Ch. 16 - Prob. 16.58PCh. 16 - Draw a stepwise mechanism for the following...Ch. 16 - Prob. 16.60PCh. 16 - Prob. 16.61PCh. 16 - Prob. 16.62PCh. 16 - Prob. 16.63PCh. 16 - Prob. 16.64PCh. 16 - 16.65 The treatment of isoprene with one...Ch. 16 - 16.66 The treatment of with forms B (molecular...Ch. 16 - Prob. 16.67PCh. 16 - Prob. 16.68PCh. 16 - Prob. 16.69PCh. 16 - Prob. 16.70PCh. 16 - Prob. 16.71PCh. 16 - Prob. 16.72PCh. 16 - Prob. 16.73PCh. 16 - Prob. 16.74PCh. 16 - Prob. 16.75P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Intramolecular Diels–Alder reactions are possible when a substrate contains both a 1,3-diene and a dienophile, as shown in the following general reaction.With this in mind, draw the product when each compound undergoes an intramolecular Diels–Alder reaction.arrow_forwardZingiberene and β-sesquiphellandrene, natural products obtained from ginger root, contain conjugated diene units. Which diene reacts faster in the Diels–Alder reaction and why?arrow_forwardRank the following in order from slowest to fastest rate of reaction in a Diels–Alder reaction with buta-1,3-dienearrow_forward
- How does bromobenzene react differently from benzyl chloride under SN1 and SN2 conditions, and why?arrow_forwardOn photolysis, A undergoes primarily Norrish II chemistry, but B undergoes primarily Norrish I chemistry. Rationalize the difference and draw the major products expected from each reaction. (note the stereochemistry in the ring structure).arrow_forwardwhat structure show the transition state for the rate determining step in the sn1 reaction with this picture?arrow_forward
- What cyclic product is formed when each decatetraene undergoes photochemical electrocyclic ring closure?arrow_forwardAlthough dehydrohalogenation occurs with anti periplanar geometry,some eliminations have syn periplanar geometry. Examine the startingmaterial and product of attached elimination, and state whether the elimination occurs with syn or anti periplanar geometry.arrow_forwardThe diene lactone shown in part (a) has one electron-donating group (¬OR) and one electron-withdrawing group(C“O). This diene lactone is sufficiently electron-rich to serve as the diene in a Diels–Alder reaction.(a) What product would you expect to form when this diene reacts with methyl acetylenecarboxylate, a strong dienophile?arrow_forward
- One step in the synthesis of dodecahedrane involves reaction of the tetraene C with dimethylacetylene dicarboxylate (D) to afford two compounds having molecular formula C16H16O4. This reaction has been called a domino Diels–Alder reaction. Identify the two products formed.arrow_forwardPredict the product for the following reaction and use molecular orbital theory to explain the stereochemical outcomearrow_forwardIdentify the reddish brown species obtained when the cyclopentadiene reacted withKOH. Why are the species and ferrocene aromatic?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License