
Concept explainers
(a)
Interpretation:
The given amine has to be named and it should be indicated whether as the ammonium salt of a primary, secondary or tertiary amine.
Concept introduction:
In chemistry Structure is the arrangement of
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
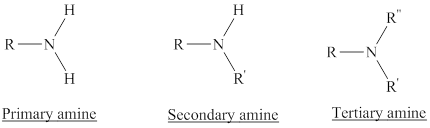
From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Ammonia is very soluble in water giving an alkaline solution which can be neutralized by acids to produce ammonium salts.
(b)
Interpretation:
The given amine has to be named and it should be indicated whether as the ammonium salt of a primary, secondary or tertiary amine.
Concept introduction:
In chemistry Structure is the arrangement of chemical bonds between atoms in a molecule, specifically which atoms are chemically bonded to what other atoms with what kind of chemical bond.
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Ammonia is very soluble in water giving an alkaline solution which can be neutralized by acids to produce ammonium salts.
(c)
Interpretation:
The structure of the given compound has to be drawn and it should be indicated whether as the ammonium salt of a primary, secondary or tertiary amine.
Concept introduction:
In chemistry Structure is the arrangement of chemical bonds between atoms in a molecule, specifically which atoms are chemically bonded to what other atoms with what kind of chemical bond.
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Ammonia is very soluble in water giving an alkaline solution which can be neutralized by acids to produce ammonium salts.
(d)
Interpretation:
The structure of the given compound has to be drawn and it should be indicated whether as the ammonium salt of a primary, secondary or tertiary amine.
Concept introduction:
In chemistry Structure is the arrangement of chemical bonds between atoms in a molecule, specifically which atoms are chemically bonded to what other atoms with what kind of chemical bond.
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Ammonia is very soluble in water giving an alkaline solution which can be neutralized by acids to produce ammonium salts.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
EP FUND.OF GENERAL,ORG...-MOD.MASTERING
- Draw the structures of the following acids:(a) 2-Ethyl-3-hydroxyhexanoic acid (b) m-Nitrobenzoic acidarrow_forwardplease helparrow_forwardIn a paragraph form, provide the experimental procedures of the development of the ring structure of α-tetralene by elimination of the water molecule will result in this product being heated in the presence of strong sulphuric acid.arrow_forward
- What chemical reaction causes the color change of diphenylamine sulfonate indicator?arrow_forwardWhat is the ionisable group of phenazopyridine? With illustrationsarrow_forward2-butanol can be formed as the only product of the Markovnikov addition of H2O to two different alkenes. In contrast, 2-octanol can be formed as the only product of the Markovnikov addition of H2O to just one alkene. To examine the difference, draw the alkene starting materials of each alcohol. Draw the bond-line (skeletal) structures of the two alkene starting materials that can be used to synthesize 2-butanol via Markovnikov hydration. Part 1 of 2 Click and drag to start drawing a structure. : ☐ ☑ ⑤arrow_forward
- (a) (S)-2-chlorobutane, draw a three-dimensional representation.arrow_forwardFor the following monosaccharide, draw the products formed when treated with each reagent. H H -OH HO H HO H H― -OH CH₂OH Part 1 of 2 H2 in the presence of a Pd catalyst. Modify the Fischer projection of the monosaccharide to represent the product. H H -ОН HO H HO H H -OH CH₂OH Part 2 of 2 Benedict's reagent. Modify the Fischer projection of the monosaccharide to represent the product. H H -OH HO -Н HO H H -OH CH₂OH ☐ : G ☐ E ☐: ☑ P Garrow_forwardFor the following monosaccharide, draw the products formed when treated with each reagent. H. HO H H OH HO H H OH Part 1 of 2 CH₂OH H₂ in the presence of a Pd catalyst. Modify the Fischer projection of the monosaccharide to represent the product. H HO -H H -OH HO -H H -OH CH₂OH Part 2 of 2 Benedict's reagent. Modify the Fischer projection of the monosaccharide to represent the product. HO H H -OH HO H H -OH CH₂OH Q:arrow_forward
- How many alkene groups are in linolenic acid? are the Alkenes cis or trans ?arrow_forwardDetermine the weight/volume of the chemicals needed to prepare the following solutions: a) 100 ml of 0.9% (w/v) saline (NaCl) b) 30 ml of 50% glycerol (v/v) c) Electrophoresis requires TAE, which is a specific mixture of Tris base, acetic acid, and EDTA. TAE is normally made as a 50X concentrated stock. Provide a recipe to make 40 ml of 50X TAE. The recipe for one liter of 50X TAE is as follows: 242g Tris base, 57.1 ml glacial acetic acid, 100 ml 0.5 M EDTAarrow_forwardExplain why pyrimidine is less basic than pyridinearrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





