
(a)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
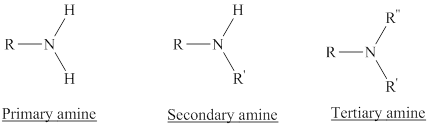
Reaction of amines and acid will give amine salt an (ammonium ion).
(b)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Reaction of amines and acid will give amine salt an (ammonium ion).
Treating an amine salt with a strong base regenerates the “parent” amine.
(c)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Reaction of amines and acid will give amine salt an (ammonium ion).
Amine are weak bases (proton
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Plus Mastering Chemistry with Pearson eText -- Access Card Package (8th Edition)
- Draw the structure of the ester formed when ethanoic acid (CH3 CO2 H) is treated with CH3 OH in the presence of H2SO4. Click and drag to start drawing a structure. ☑arrow_forwardDraw a structure for the compound, C3H5Br, that fits the following 1H NMR data: δ 2.32 (3H, singlet) δ 5.35 (1H, broad singlet) δ 5.54 (1H, broad singlet)arrow_forwardDraw dipeptide Arg-Thr at pH 6.0. Circle the six atoms that are restricted in one plane. Calculate its approximate pI assuming pKa for NH3+ is 10.0, pKa for COOH is 3.0, PKR for Arg is 12.0arrow_forward
- Convert the following Fischer structures into cyclic Haworth structures:arrow_forwardDraw structural formulas for all of the following. Q.) Alcohols with the molecular formula C4H10Oarrow_forwardDraw the stereoisomers of the following amino acids. Indicate pairs of enantiomers and pairs of diastereomers. leucineCH3CHCH2 CHCOO−CH3 +NH3 isoleucineCH3CH2CH CHCOO−CH3arrow_forward
- Draw the structure of the product that forms when the carbonyl compound shown is treated with K₂ Cг2 О7. If no reaction occurs, draw the structure of the organic starting material (reactant). CH3(CH2), (CO)CH (CH3)2 Click and drag to start drawing a structure. : ☐arrow_forwardPlease step by step answer and don't use ASI Answer pleasearrow_forwardIdentify the conjugate acid-base pairs in the following reactions: HNO2(aq) + H2O(l) → NO2 – (aq) + H3O+(aq) _______ ______ _________ ________ CH3NH2 + H2O(l) → CH3NH3+ + OH – _______ ________ ________ _________arrow_forward
- Consider the alkene 5,5-dimethyl-3-octene for the following problems. Part 1 of 2 Draw a skeletal structure for the cis isomer of 5,5-dimethyl-3-octene. Click and drag to start drawing a structure. ☑ :୮arrow_forwardHere are the SALCS group orbitals of the two F atoms in XeF2. Using the D2h character table, assign the symmetry symbol for the labeled SALCS #3 and #8. #1 #2 ・ O O 800 6 F #3 St 5. #5 #7 8 O #4 #6 2·脚·· · #8 8 F Xe F 8 # 3 is Ag and #8 is B3g O #3 is Blu and #8 is B2g O #3 is Ag and #8 is B2u O#3 is B1g and #8 is B1u # 3 is Blu and #8 is B3g Farrow_forwardThe ionization of p-nitrophenol is shown below (pKa = 7.0): a. Identify the weak acid and conjugate base. b. At pH 7, what are the relative concentrations of ionized and un-ionized p-nitrophenol? c. If enough concentrated hydrochloric acid is added to a solution of p-nitrophenol to lower the pH from 7 to 5, what will happen to the relative concentrations of the ionized and un-ionized forms? d. Ionized p-nitrophenol has a yellow color, while the un-ionized form is colorless. The yellow color can be measured using a spectrophotometer at 400nm. In order to determine the total amount of p-nitrophenol in a solution, would you perform the spectrophotometer reading at an acidic or basic pH? Clearly explain why? e. A solution of p-nitrophenol at pH 7.95 was found to have an A400 of 0.255 . What is the total concentration (in µM) of p-nitrophenol (ionized plus un-ionized) in the solution? The molar extinction coefficient of p-nitrophenol is 18,500 M-1cm-1 and the pKa is 7.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





