
(a)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
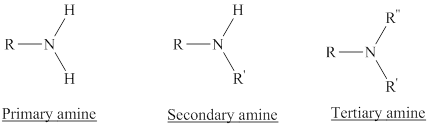
Reaction of amines and acid will give amine salt an (ammonium ion).
(b)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Reaction of amines and acid will give amine salt an (ammonium ion).
Treating an amine salt with a strong base regenerates the “parent” amine.
(c)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Reaction of amines and acid will give amine salt an (ammonium ion).
Amine are weak bases (proton
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
FUND. GEN. ORG&BIO CHEM PKG
- Identify the acid on the left and its conjugate base on the right in the following equations:(a) HOCl + H2O ↔ H3O+ + OCl-(b) HONH2 + H2O ↔ HONH3+ + OH-(c) NH4+ + H2O ↔ NH3 + H3O+(d) 2HCO3-2 ↔ H2CO3 + CO3-2 (e) PO4-3 + H2PO4- ↔ 2HPO4-2arrow_forwardIdentify the names of the following molecules.arrow_forwardFor A, B, C, D, E, F, identify the circled functional groups and linkages in the compound in the picture.arrow_forward
- With the given structures, arrange the following compounds in order of decreasing reactivity towards hydrolysisarrow_forwardBalance the following equation, and tell how many moles of nickel will reactwith 9.81 mol of hydrochloric acid.arrow_forwardIndicate whether each of the following statement about enantiomers is True or False. a. Enantiomers always have the same molecular formula. b. Enantiomers always have the same structural formula. c. Enantiomers are always nonsupetrimposable mirror images of each other. d. Enantiomers always differ in handedness.arrow_forward
- Compute the formal charge (FC) on each atom in the following structures [H2CNH2]+arrow_forwardConsider the intermolecular forces present in a pure sample of each of the following compounds: CH₃CH₂OH and CH₃COCH₃. Identify the intermolecular forces that these compounds have in common.arrow_forwardWhat is the empirical formula for C3H6O3? C3H6O3 C6H12O6 CH2O None of thesearrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





