
(a)
Interpretation:
The given reactions are final steps in one industrial synthesis of vitamin A acetate.
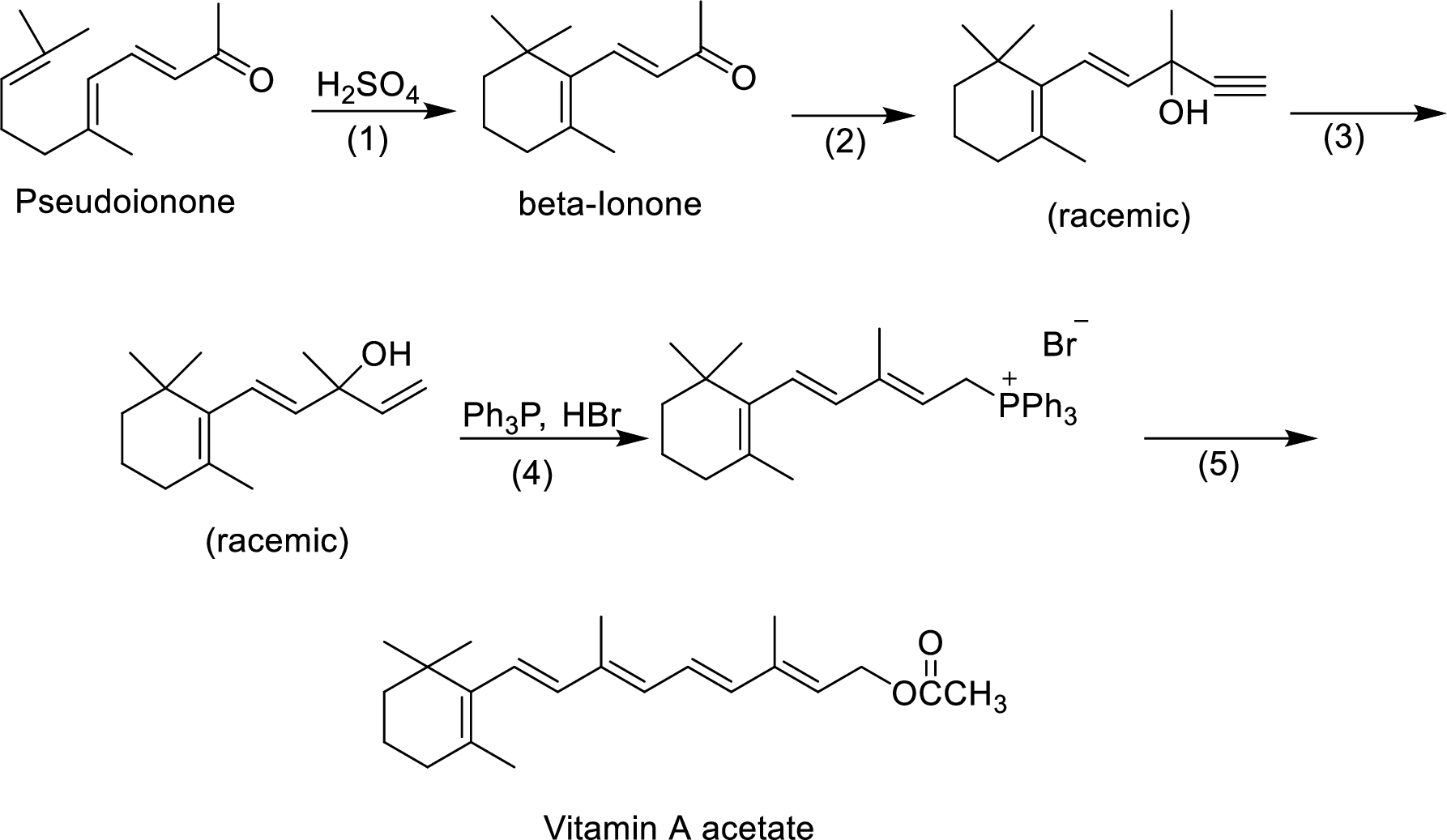
The mechanism has to be proposed for the acid-catalyzed cyclization in step-1.
Concept Introduction:
The ring cyclization occurs when an acyclic molecule reacts with acid. by the abstraction of proton from the acid molecule, internal bond shifting occurs to form the cyclization product.
(b)
Interpretation:
The given reactions are final steps in one industrial synthesis of vitamin A acetate.
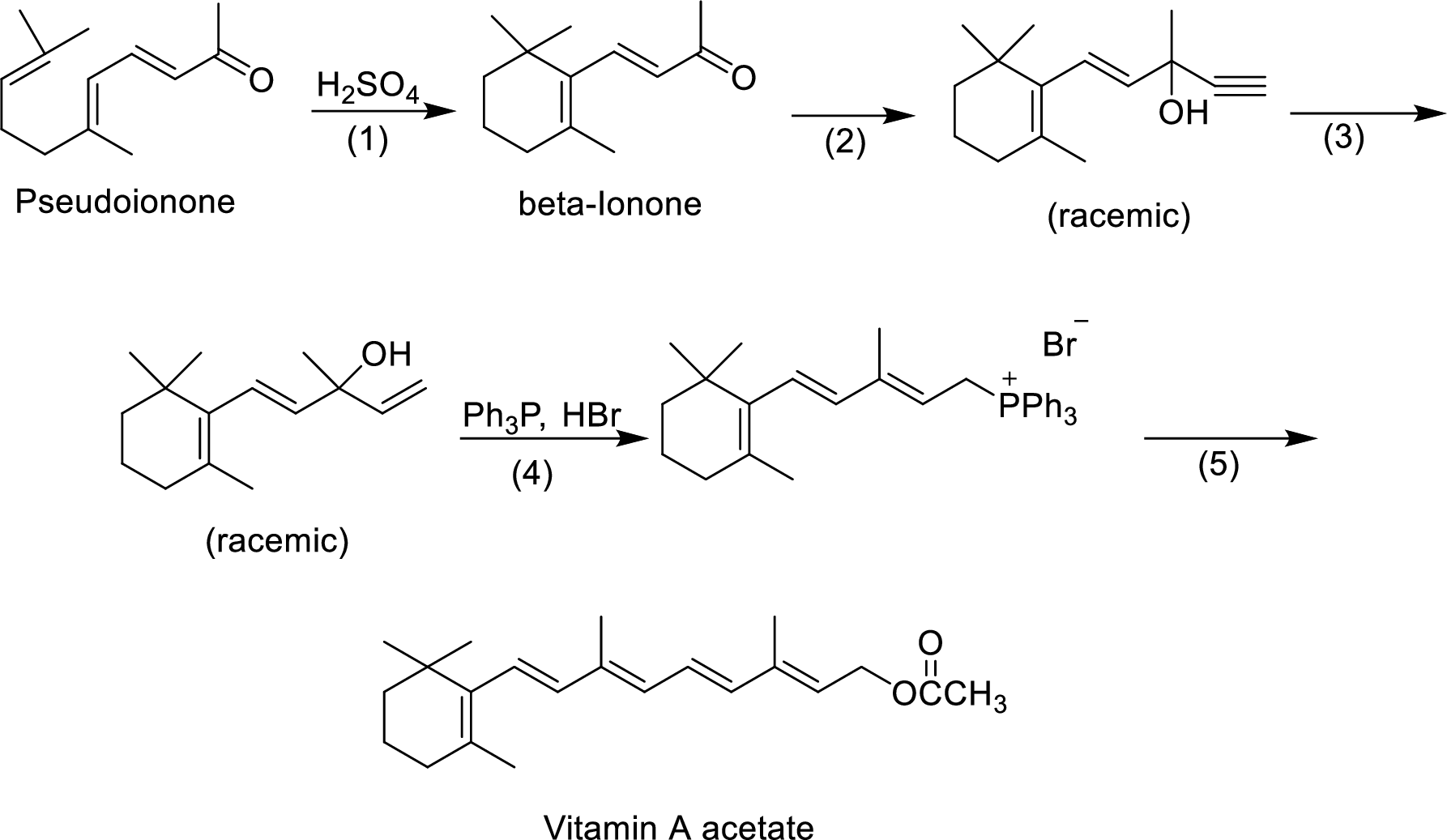
The reagents has to be given for the reaction involved in step-2.
(c)
Interpretation:
The given reactions are final steps in one industrial synthesis of vitamin A acetate.
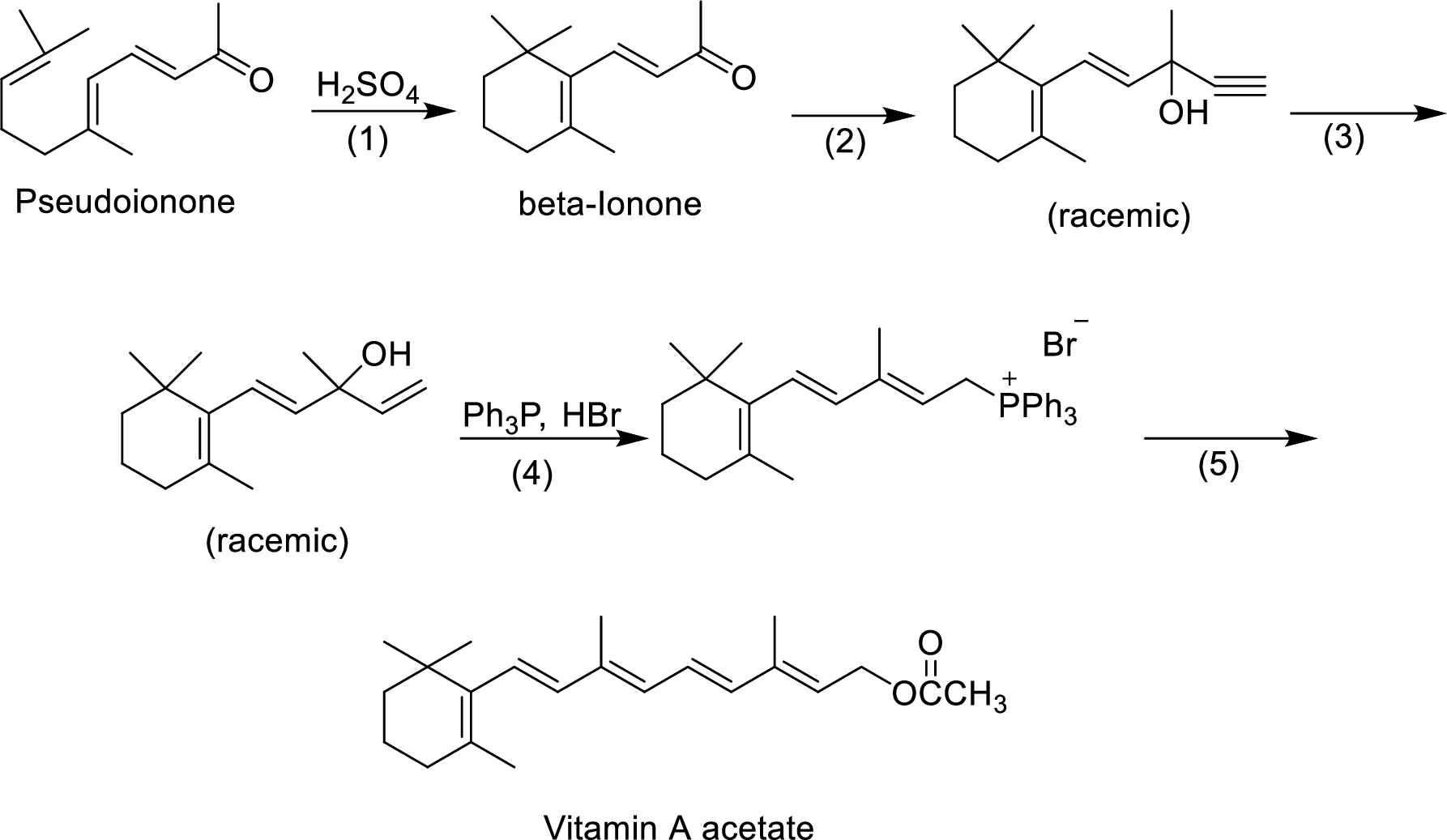
The mechanism has to be proposed for the formation of phosphonium salt in step-4.
(d)
Interpretation:
The given reactions are final steps in one industrial synthesis of vitamin A acetate.
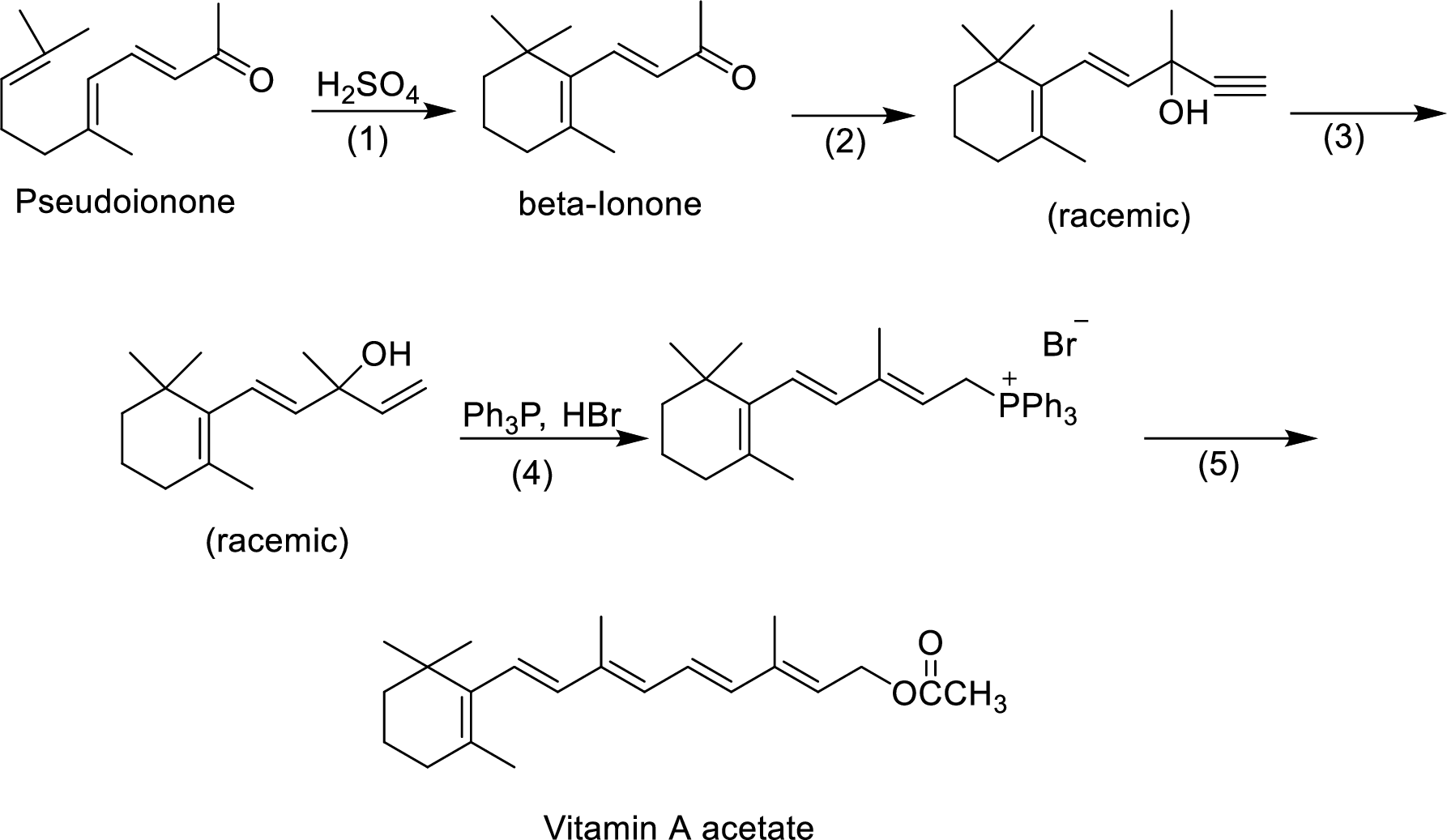
The reagents that are involved in the reaction of step-3 has to be proposed.
(e)
Interpretation:
The given reactions are final steps in one industrial synthesis of vitamin A acetate.
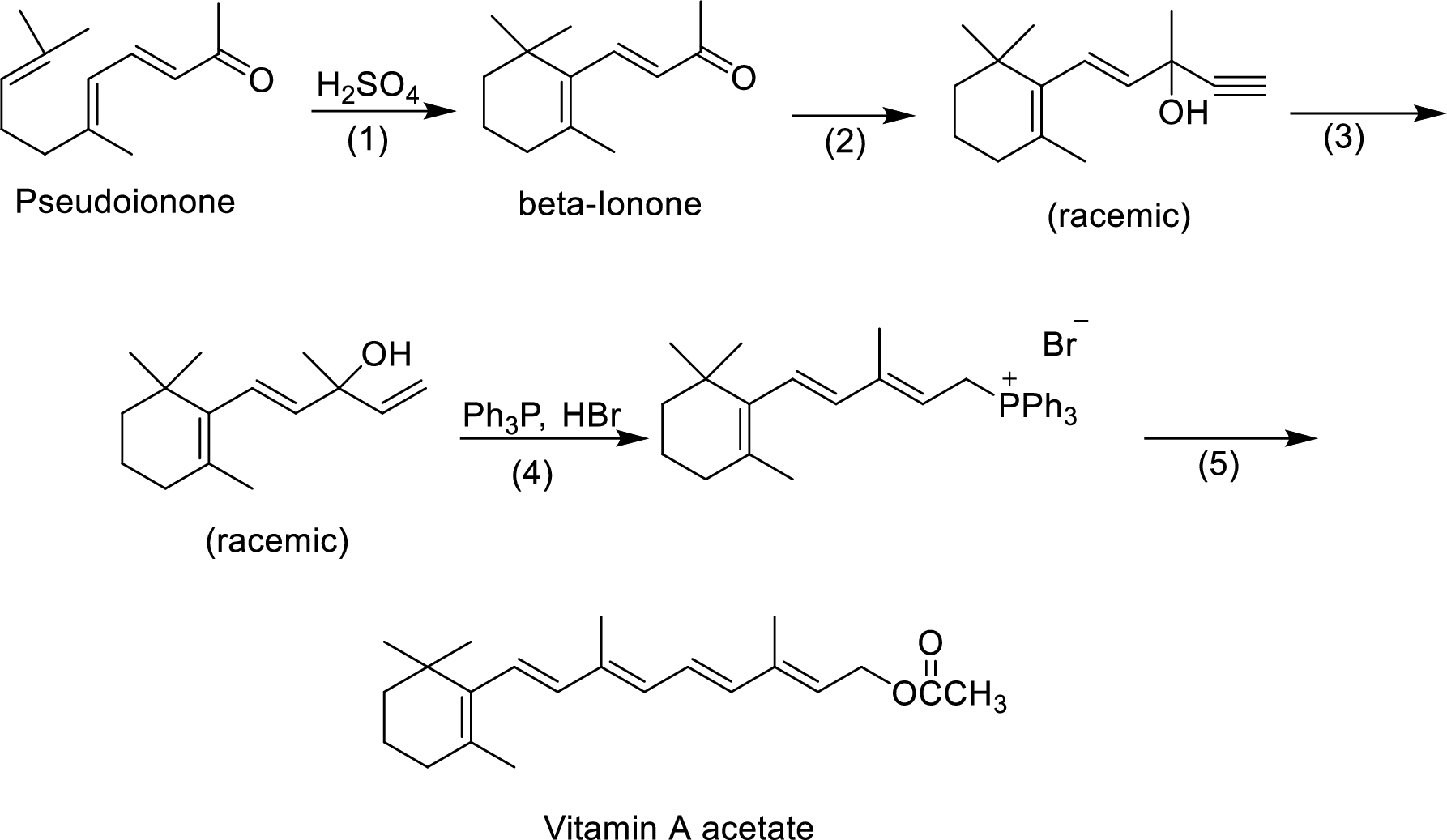
Wittig reaction of step-5 has to be shown.
Concept Introduction:
Wittig reaction:
The reaction of carbonyl compound with the ylide of phosphonium salt to give an
Trending nowThis is a popular solution!

Chapter 16 Solutions
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- (b) Answer ALL parts (i)-(iv) of this question. The scheme below outlines a synthesis of a vitamin A analogue. (i) Suggest reagents for the conversion of 1 into 2 and write a mechanism for this reaction; (ii) Suggest a reagent for the conversion of 3 into 4; (iii) Propose a mechanism for the conversion of 4 into 5; (iv) Suggest reagents for the conversion of 5 into 6 and provide a mechanism for this transformation. reagents? 2 reagents? PPH3; HBr ÓH 3 4 PPH3 Br reagents?arrow_forwarda)Write the SN2 reaction mechanism between iodobutane with sodium hydroxide, NaOH. b)Predict the products and show the SN1 reaction mechanism that occurs with 2-iodo-2- methylpropane in aqueous sodium hydroxide, NaOH.arrow_forwardPredict the intermediates and final product. Provide a rational mechanism.arrow_forward
- Please explain the synthesis. Identify SN1, SN2, E1, E2, nucleophiles and ekectrophiles.arrow_forwardb) Listed below are several hypothetical nucleophilic substitution reactions. None is synthetically useful because the product indicated is not formed at an appreciable rate. In each case provide an explanation for the failure of the reaction to take place as indicated. OMe HO + OMe + OH HO + CH; OHarrow_forward7 Compound 5 can be made from compound 1 in the following reaction sequence. Br OH step 1 step 2 + NaOH + P 1 R 2 OH OH step 5 + dil. H₂SO4 I - S 5 HOOC 4 NC (a) Suggest a reagent P that can be used in step 1. (b) What name would you give for the reaction occurring in step 1? (c) Suggest a name for compound 2. Name: (d) Name the inorganic product R that is also formed in step 2. (e) Suggest the name of the mechanism for step 2. (f) Draw a fully labelled reaction mechanism for step 2. step 4 + HCN step 3arrow_forward
- Propose a synthesis of molecule B from molecule A. For each step, indicate the necessary reagents.arrow_forwardAddition of one equivalent of ammonia to 1-bromoheptane gives a mixture of heptan-1-amine,some dialkylamine, some trialkylamine, and even some tetraalkylammonium bromide.(a) Give a mechanism to show how this reaction takes place, as far as the dialkylamine.(b) How would you modify the procedure to get an acceptable yield of heptan-1-amine?arrow_forward(a) Decide whether the reaction below will proceed via an E1 or E2 and write a step-by-step mechanism. (b) Draw structural formula from the major organic product only.arrow_forward
- Predict the product of the following reactions. (a) 1. NaOEt, EtOH, heat 2. H3O+ quench (b) Pho O O OEt OMe 1. NaOMe, MeOH, heat 2. H3O+ quencharrow_forwardSuggest a suitable mechanism for this multi-step reaction, and give products A and Barrow_forward(b) Answer the following questions based on the compounds below. Jawab soalan berikut berdasarkan kepada sebatian di bawah. CI CI A в (i) Which compound has the higher boiling point? Explain. Sebatian manakah mempunyai takat didih yang lebih tinggi? Terangkan. (ii) Draw the SN2 mechanism for the reaction of compound A with sodium hydroxide, NaOH. Lukis mekanisma Sn2 bagi tindak balas antara sebatian A dengan natrium hidroksida, NaOH.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
