
(a)
Interpretation:
Show how the given
Concept introduction:
The
Wittig reaction is a reaction in which a nucleophilic elimination occurs after nucleophilic addition reaction. In this reaction an aldehyde or ketone reacts with a triphenyl phosphonium ylide (Wittig reagent) to give an alkene and triphenylphosphine oxide.
The reaction can be represented as shown below,
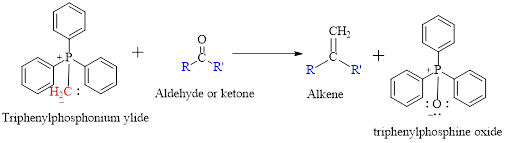
(b)
Interpretation:
Show how the given alkene can be synthesized by a Wittig reaction.
Concept introduction:
The functional group in the aldehydes and Ketones are carbonyl group.
Wittig reaction is a reaction in which a nucleophilic elimination occurs after nucleophilic addition reaction. In this reaction an aldehyde or ketone reacts with a triphenyl phosphonium ylide (Wittig reagent) to give an alkene and triphenylphosphine oxide.
The reaction can be represented as shown below,

Trending nowThis is a popular solution!

Chapter 16 Solutions
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- For the following reaction scheme, match the correct reagent to each reaction (A, B, C, D and E).arrow_forwardDraw the products of each reaction, and state whether the reaction is fasteror slower than a similar reaction with benzene.arrow_forwardDraw the products of each reaction, and indicate the stereochemistry where appropriate. With explanation on how did you get the answer.arrow_forward
- Treatment of alkenes A and B with HBr gives the same alkyl halide C. Draw a mechanism for each reaction, including all reasonable resonance structures for any intermediate.arrow_forwardDraw the products of each reaction, and indicate the stereochemistry where appropriate.arrow_forwardDraw a stepwise, detailed mechanism for the following reaction. CH3NH2 N-CH3 CH,NH, Cr (excess)arrow_forward
- Please transform and give starting material or product. Show stereochemistry and mechanism as needed.arrow_forwardDraw the products of each reaction, including stereochemistry.arrow_forwardShow how to synthesize the following product as the major product using any reactants/reagents and number of steps. Be sure to list each step with all reactants/reagents/conditions required. (Do not use hydrogenation reactions)arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
