
(a)
Interpretation:
The structural formulas for the products of hydrolysis of the given acetal in aqueous acid has to be drawn.
Concept introduction:
Acetals are a group of organic compound that contain two alkoxy (-OR) groups on the same carbon atom.
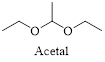
Acetals can be hydrolysed by addition of dilute acid. The excess of water drives the equilibrium toward the formation of the ketone or
(b)
Interpretation:
The structural formulas for the products of hydrolysis of the given acetal in aqueous acid has to be drawn.
Concept introduction:
Acetals are a group of organic compound that contain two alkoxy (-OR) groups on the same carbon atom.

Acetals can be hydrolysed by addition of dilute acid. The excess of water drives the equilibrium toward the formation of the ketone or aldehyde and two molecules of alcohol or one molecule of diol.
(c)
Interpretation:
The structural formulas for the products of hydrolysis of the given acetal in aqueous acid has to be drawn.
Concept introduction:
Acetals are a group of organic compound that contain two alkoxy (-OR) groups on the same carbon atom.

Acetals can be hydrolysed by addition of dilute acid. The excess of water drives the equilibrium toward the formation of the ketone or aldehyde and two molecules of alcohol or one molecule of diol.
Trending nowThis is a popular solution!

Chapter 16 Solutions
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- Draw structural formulas for the following entities. a. Propanoate ion b. Sodium propanoate c. Acetate ion d. Sodium acetatearrow_forwardWhat is the oxidation product of this compound? propanone ethanoic acid acetic acid propanoic acidarrow_forwardWhen trans-2-chloro-1-cyclohexanol is treated with a base, cyclohexene oxide is the product. However, when cis-2-chloro-1-cyclohexanol is treated with a base, the product is cyclohexanone A.Write the equation for the reaction between trans-2-chloro-1-cyclohexanol and the base to yield the cyclohexene oxide B.Why doesn’t the cis isomer yield the oxide? C.Write the mechanism for each of the two reactions.arrow_forward
- 1. What is the role of the acetic acid in the oxidation of Cyclohexanol to Cyclohexanone? Write the balanced chemical reaction between acetic acid and sodium hypochlorite.2. How do you neutralize the acetic acid regenerated in the reaction? Write the balanced chemical reaction.arrow_forwardList the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forwardAldehydes and ketones react with one molecule of an alcohol to form compounds called hemiacetals, in which there is one hydroxyl group and one ether-like group. Reaction of a hemiacetal with a second molecule of alcohol gives an acetal and a molecule of water. We study this reaction in Chapter 16. Draw structural formulas for the hemiacetal and acetal formed from these reagents. The stoichiometry of each reaction is given in the problem.arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





