
Acetic acid, CH3CO2H, can form a dimer, (CH3CO2H)2, in the gas phase..
The dimer is held together by to hydrogen bonds with a total strength of 66.5 kJ per mole of dimer.
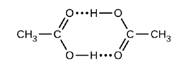
At 25 °C, the equilibrium constant for the dimerization is
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Chemistry by OpenStax (2015-05-04)
Additional Science Textbook Solutions
College Physics
Organic Chemistry (8th Edition)
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry: The Central Science (14th Edition)
Introductory Chemistry (5th Edition) (Standalone Book)
- Describe a nonchemical system that is in equilibrium, and explain how the principles of equilibrium apply to the system.arrow_forwardAt 500C, k for the for the formation of ammonia from nitrogen and hydrogen gases is 1.5105. N2(g)+3H2(g)2NH3(g)Calculate the equilibrium partial pressure of hydrogen if the equilibrium partial pressures of ammonia and nitrogen are 0.015 atm and 1.2 atm, respectively.arrow_forwardHydrogen gas and iodine gas react to form hydrogen iodide. If 0.500 mol H2 and 1.00 mol I2 are placed in a closed 10.0-L vessel, what is the mole fraction of HI in the mixture when equilibrium is reached at 205C? Use data from Appendix C and any reasonable approximations to obtain K.arrow_forward
- Consider the reaction 2HI(g)H2(g)+I2(g)At 500C a flask initially has all three gases, each at a partial pressure of 0.200 atm. When equilibrium is established, the partial pressure of HI is determined to be 0.48 atm. What is G for the reaction at 500C?arrow_forwardRed phosphorus is formed by heating white phosphorus. Calculate the temperature at which the two forms are at equilibrium, given white P: H f =0.00 kJ/mol; S =41.09 J/mol K red P: H f =17.6 kJ/mol; S =22.80 J/mol Karrow_forwardThe following data were collected for a system at equilibrium at 140°C. Calculate the equilibrium constant for the reaction, 3 H2(g) + N2(g) 5=^ 2 NHt(g) at this temperature. [H2] = 0.10 mol L_1, [NJ = 1.1 mol L"1, [NHJ = 3.6 X 10"-mol L'1arrow_forward
- . Many sugars undergo a process called mutarotation, in which the sugar molecules interconvert between two isomeric forms, finally reaching an equilibrium between them. This is true for the simple sugar glucose, C6H12O6, which exists in solution in isomeric forms called alpha-glucose and beta-glucose. If a solution of glucose at a certain temperature is analyzed, and it is found that the concentration of alpha-glucose is twice the concentration of beta-glucose, what is the value of K for the inter-conversion reaction?arrow_forward12.101 An engineer working on a design to extract petroleum from a deep thermal reservoir wishes to capture toxic hydrogen sulfide gases present by reaction with aqueous iron(II) nitrate to form solid iron(II) sulfide. (a) Write the chemical equation for this process, assuming that it reaches equilibrium. (b) What is the equilibrium constant expression for this system? (c) How can the process be manipulated so that it does not reach equilibrium, allowing the continuous removal of hydrogen sulfide?arrow_forwardLexan is a plastic used to make compact discs, eyeglass lenses, and bullet-proof glass. One of the compounds used to make Lexan is phosgene (COCl2), an extremely poisonous gas. Phosgene decomposes by the reaction COCl2(g)CO(g)+Cl2(g) for which Kp 6.8 109 at 100C. If pure phosgene at an initial pressure of 1.0 atm decomposes, calculate the equilibrium pressures of all species.arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





