
ORGANIC CHEM ALC PKG
8th Edition
ISBN: 9780134595634
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 69P
How many signals would the product of the following reaction show in
- a. its 1H NMR spectrum?
- b. its 13C NMR spectrum?
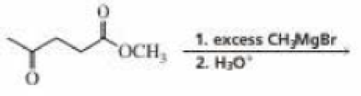
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Grinard Reaction with Aldehyde:
(a) What peaks in the NMR spectrum most clearly demonstrate the presence of the predicted product ,1-(4-methoxypenyl)-methylpropan-1-ol?
(b) If the product had unreacted aldehyde remaining, what NMR peak would indicate the presence of this contaminant?
Which does the 1H NMR represent: the starting material or the product of the reaction?
What product did you obtain from the competitive nitration of methyl benzoate? a. Which particular IR absorptions led you to this conclusion? b. How can the NMR signals in the aromatic region be used to confirm the identity of your product?
Chapter 16 Solutions
ORGANIC CHEM ALC PKG
Ch. 16.1 - Give two names for each of the following:Ch. 16.1 - Prob. 2PCh. 16.1 - Name the following:Ch. 16.2 - Prob. 4PCh. 16.4 - What products are formed when the following...Ch. 16.4 - We saw on the previous page that...Ch. 16.4 - a. How many stereoisomers are obtained from the...Ch. 16.4 - Prob. 9PCh. 16.4 - Write the mechanism for the reaction of acetyl...Ch. 16.4 - Prob. 11P
Ch. 16.4 - Show how the following compounds can be...Ch. 16.4 - Prob. 13PCh. 16.4 - Prob. 14PCh. 16.4 - In the mechanism for cyanohydrin formation, why is...Ch. 16.4 - Prob. 16PCh. 16.4 - Prob. 17PCh. 16.4 - Show two ways to convert an alkyl halide into a...Ch. 16.5 - Prob. 20PCh. 16.5 - Prob. 21PCh. 16.5 - Prob. 22PCh. 16.5 - Prob. 23PCh. 16.6 - Prob. 24PCh. 16.7 - What reducing agents should be used to obtain the...Ch. 16.7 - Prob. 26PCh. 16.8 - Prob. 27PCh. 16.8 - Prob. 28PCh. 16.8 - Prob. 29PCh. 16.8 - The pKa of protonated acetone is about 7.5. and...Ch. 16.8 - Prob. 31PCh. 16.8 - Prob. 32PCh. 16.8 - Prob. 33PCh. 16.8 - Excess ammonia must be used when a primary amine...Ch. 16.8 - The compounds commonly known as amino acids are...Ch. 16.9 - Hydration of an aldehyde is also catalyzed by...Ch. 16.9 - Which ketone forms the most hydrate in an aqueous...Ch. 16.9 - When trichloroacetaldehyde is dissolved in water,...Ch. 16.9 - Which of the following are a. hermiacetals? b....Ch. 16.9 - Prob. 40PCh. 16.9 - Explain why an acetal can be isolated but most...Ch. 16.10 - Prob. 42PCh. 16.10 - Prob. 43PCh. 16.10 - What products would be formed from the proceedings...Ch. 16.10 - a. In a six-step synthesis, what is the yield of...Ch. 16.10 - Show how each of the following compounds could be...Ch. 16.12 - Prob. 47PCh. 16.13 - Prob. 49PCh. 16.14 - Prob. 50PCh. 16.15 - Prob. 51PCh. 16.16 - Prob. 52PCh. 16 - Draw the structure for each of the following: a....Ch. 16 - Prob. 54PCh. 16 - Prob. 55PCh. 16 - Prob. 56PCh. 16 - a. Show the reagents required to form the primary...Ch. 16 - Prob. 58PCh. 16 - Prob. 59PCh. 16 - Using cyclohexanone as the starting material,...Ch. 16 - Propose a mechanism for each of the following...Ch. 16 - Show how each of the following compounds can be...Ch. 16 - Fill in the boxes:Ch. 16 - Prob. 64PCh. 16 - Identify A through O:Ch. 16 - Prob. 66PCh. 16 - Prob. 67PCh. 16 - Prob. 68PCh. 16 - How many signals would the product of the...Ch. 16 - Prob. 70PCh. 16 - Prob. 71PCh. 16 - Prob. 72PCh. 16 - Prob. 73PCh. 16 - Prob. 74PCh. 16 - Prob. 75PCh. 16 - Prob. 76PCh. 16 - Prob. 77PCh. 16 - Prob. 78PCh. 16 - Draw structure for A-D for each of the following:Ch. 16 - Prob. 80PCh. 16 - a. Propose a mechanism for the following reaction:...Ch. 16 - Prob. 82PCh. 16 - A compound gives the following IR spectrum. Upon...Ch. 16 - How can be following compounds be prepared from...Ch. 16 - Prob. 85PCh. 16 - Prob. 86PCh. 16 - Prob. 87PCh. 16 - In the presence of an acid catalyst, acetaldehyde...Ch. 16 - Prob. 89PCh. 16 - Prob. 90PCh. 16 - Prob. 91PCh. 16 - A compound reacts with methylmagnesium bromide...Ch. 16 - Show how each of the following compounds can be...Ch. 16 - Prob. 94PCh. 16 - The pKa values of the carboxylic acid groups of...Ch. 16 - The Baylis-Hillman reaction is a DABCO...Ch. 16 - Prob. 97PCh. 16 - Prob. 98P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For the product, triphenylmethanol, what peaks should we see in 1) 1H NMR 2) 13C NMR, and 3) IR spectra?arrow_forward5. How does the proton (¹H) NMR of the methyl benzoate reagent compare to the methyl 3-nitrobenzoate product? Include (or illustrate) the NMR plots and peak assignments for each.arrow_forwardDraw the NMR spectrum expected from ethanol that has been shaken with a drop of D2Oarrow_forward
- What is the 1H NMR data (chemical shift, integration, multiplicity) of p-t-butyl phenol. Label the hydrogens that are being reported. Thank you for the helparrow_forward1) what is the struture of B, mechanism for its formation and assigment of the IR and 13C spectra 2) what is the struture of C, mechanism for its formation and assigment of the 1H NMR spectra 3) what is the mechanism of C to D in step 4arrow_forwardHow does the proton (1H) NMR of the methyl benzoate reagent compare to the methyl 3-nitrobenzoate product? Include (or illustrate) the NMR plots and peak assignments for each please type out so i can read clearlyarrow_forward
- What is the purpose for all of the following ? Carry out an electrophilic aromatic substitution reaction Prepare methyl m-nitrobenzoate by the nitration of methyl benzoate Verify the identity of a product through infrared spectroscopyarrow_forwardIn an Electrophilic Aromatic Reaction explain: 1. why the –NHCOCH3 group in acetanilide is only moderately activating while the -NH2 group in aniline is strongly activating? 2.Also explain why in a Electrophilic Aromatic Reaction why potassium bromate is used in this reaction instead of Br2 directly?arrow_forwardExplain why the cyclopentadienide anion A gives only one signal in its 13C NMR spectrum.arrow_forward
- i need help with predicting the Ir spec for the final product i also need to make it look like the example with the table and the 1h-nmr digram i made an example of how it needs to look like the lab called Electrophilic Aromatic Substitution: Friedel-CraftsAlkylationarrow_forwardCreat a 1H NMR data summary using ACS guidelines for Methyl 4-iodobenzoate based on the information given images given:arrow_forward: Encircle the structure corresponding to the given 1H and 13C NMR spectraarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY