
Concept explainers
a.
Interpretation:
The conversion of N-methylbenzylamine to given compound has to be explained.
Concept introduction:
The molecular formula of N-methylbenzamide is
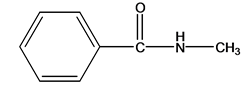
b.
Interpretation:
The conversion of N-methylbenzylamine to benzoic acid has to be explained.
Concept introduction:
The molecular formula of N-methylbenzamide is

c.
Interpretation:
The conversion of N-methylbenzylamine to methyl benzoate has to be explained.
Concept introduction:
The molecular formula of N-methylbenzamide is

d.
Interpretation:
The conversion of N-methylbenzylamine to benzyl alcohol has to be explained.
Concept introduction:
The molecular formula of N-methylbenzamide is

Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
ORGANIC CHEM ALC PKG
- Why do you wash the dichloromethane solution of your reductive amination product with sodium bicarbonate, rather than dilute aqueous HCl? a) Sodium bicarbonate is a good method of removing aldehydes from organic solvent.b) The amine product will be protonated by acid and remain in the aqueous layer as a salt.c) Sodium bicarbonate transfers the amine starting material into the aqueous layer.d) Sodium bicarbonate reacts with leftover NaBH(OAc)3 and removes it from the mixture.arrow_forwarda. Give at least three characteristics of dichloromethane that makes it a good extracting solvent for the alkaloid.b. Why is it necessary to remove a stopper from a separatory funnel when the liquid is being drained from it through a stopcock?c. What are emulsions? Why do they form during extractions? How is the formation of an emulsion minimized? How are emulsions removed?arrow_forwardWhy will ninhydrin reagent cause a blue-purple stain on the skin of your fingers when spilled. (Hint! Which group will it react with on your fingers?)arrow_forward
- What is the most likely organic product of the exhaustive hydrolysis of PhCN? A. benzoic acid B. benzamide C. benzylamine D. benzenearrow_forwardWhich of these statements is NOT true about the reaction of amines with a cyclic anhydride like phthalic anhydride? a. Dimethylamine will give a cyclic imide product. b. Aniline will give a cyclic imide product. c. Triethylamine will give a cyclic imide product. d. The reaction undergoes a nucleophilic acyl substitution mechanism.arrow_forward1. Explain, in 1 – 3 sentences, the difference in the reaction of aniline with H2SO4 and HCl. 2. Write the chemical equation for the saponification reaction. 3. Write the chemical equation for the synthesis of detergent. 4. Explain the cleaning property of soaps and synthetic detergents based on their emulsifying action. 5. Between soap and synthetic detergent, which is a better cleaning agent in hard water? Explain in 1 – 3 sentences.arrow_forward
- How to Preparation of 1-pentanamine from butanolarrow_forwardWhat is the byproduct of the following reaction, not the phenylthiohydantoin?arrow_forwardWhat must be the starting material to carry out the following reaction? a) butanamide b) 4-aminobutanol c) 1-amino-2-butanone d) propanamide e) butanenitrilearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





