
Concept explainers
a.
Interpretation:
Thiols can be prepared from the reaction of thiourea with an
Concept introduction:
Organosulfur compound that contains a carbon –bonded sulfhydry group are called thiols. It has strong odour. The –SH froup is called a mercapto group. Named by adding the suffix –thiol to the
For example, the structure of 2-methyl propane 2-thiol.
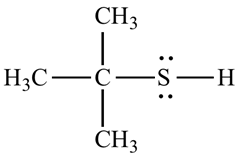
b.
Interpretation:
Thiols can be prepared from the reaction of thiourea with an alkyl halide , followed by hydroxide –io-promoted hydrolysis. What will be formed if the alkyl halide employed is pentyl bromide.
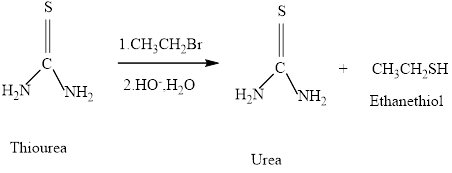
Concept introduction:
Organosulfur compound that contains a carbon –bonded sulfhydry group are called thiols. It has strong odour. The –SH froup is called a mercapto group. Named by adding the suffix –thiol to the alkane name. They are commonly prepared by an
For example, the structure of 2-methyl propane 2-thiol.
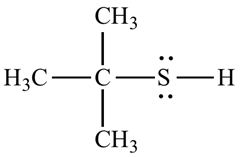
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
ORGANIC CHEM ALC PKG
- Thiols can be prepared from the reaction of thiourea with an alkyl halide, followed by hydroxide-ion-promoted hydrolysis. a. Propose a mechanism for the reaction. b. What thiol will be formed if the alkyl halide employed is pentyl bromide?arrow_forwardThiols can be prepared from the reaction of thiourea with an alkyl halide, followed by hydroxide-ion-promoted hydrolysis.a. Propose a mechanism for the reaction.b. What thiol will be formed if the alkyl halide employed is pentyl bromide?arrow_forwardHow Wittig reagents are synthesized by a two-step procedure ?arrow_forward
- Reactions carried out: a) aniline with tosyl chlorode b) N-methylaniline with tosyl choride c) aniline with nitrous acid followed by reaction with B-naphtholarrow_forward(a) Draw resonance structures for the carbocation formed after loss of a leaving group from phytyl diphosphate.(b) Draw the two-step mechanism for Friedel–Crafts alkylation of 1,2-dihydroxynaphthoic acid with this carbocation to form X.arrow_forwardFriedel-Crafts alkylations usually invariably result in a mixture of products, but Friedel acylations never do. Why? a) none of these b) alkylations require catalyst c) benzene with alkyl groups are more reactive than acylbenzenes d) the reagents used in alkylations are strongerarrow_forward
- The reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols. a. Propose a mechanism for the Ritter reaction.b. Why does the Ritter reaction not work with primary alcohols?c. How does the Ritter reaction differ from the acid-catalyzed hydrolysis of a nitrile to form an amide?arrow_forwarda. Draw the structures of all the elimination products. b. The reaction occurs through an E2 mechanism. Using one of your products writethe mechanism of the reaction using curved arrows. c. Is this reaction regioselective? Explain your answer. d. Explain why the reaction is stereoselective.arrow_forwardComplete the reaction and draw: Acetophenone reacts with phenylmagnesiumbromide in presence of H2O and then product is treated with PCl5arrow_forward
- Propose a route to dipropylzinc from propyl bromide and any necessary organic or inorganic reagents.arrow_forwardThe reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols. a. Why does the Ritter reaction not work with primary alcohols? b. Provide an explanation for why an amide is less susceptible to nucleophilic attack than its corresponding ester.arrow_forwardDraw the product formed when pentanal (CH3CH2CH2CH2CHO) is treatedwith following reagent. With some reagents, no reaction occurs. Na2Cr2O7, H2SO4, H2Oarrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

