
Concepts of Genetics (11th Edition)
11th Edition
ISBN: 9780321948915
Author: William S. Klug, Michael R. Cummings, Charlotte A. Spencer, Michael A. Palladino
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 6PDQ
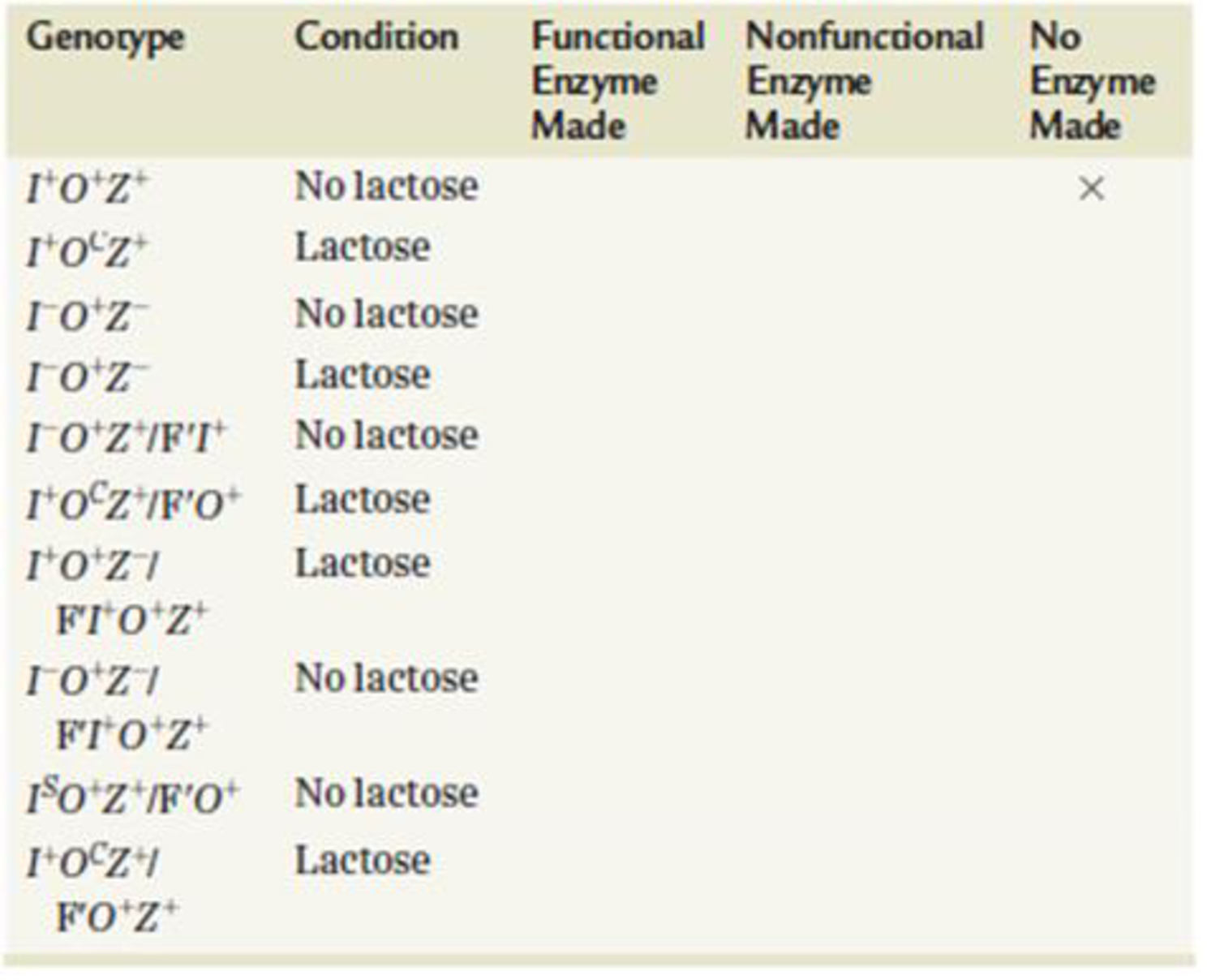
For the genotypes and conditions (lactose present or absent) shown in the following table, predict whether functional enzymes, nonfunctional enzymes, or no enzymes are made.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the α-l-glucopyranose sugars using Haworth projections:
The enzyme glucose oxidase isolated from the mold Penicillium notatum catalyzes the oxidation of β-Dglucose to D-glucono-δ-lactone. This enzyme is highly specific for the β anomer of glucose and does not affect the α anomer. In spite of this specificity, the reaction catalyzed byglucose oxidase is commonly used in a clinical assay for total blood glucose—that is, for solutions consisting of a mixture of β- and α-D-glucose. What are the circumstances required to make this possible? Aside from allowing the detection of smaller quantities of glucose, what advantage does glucose oxidase offer over Fehling’s reagent for measuring blood glucose?
The enzyme glucose oxidase isolated from the mold Penicillium notatum catalyzes the oxidation of
B-D-glucose to D-glucono-6-lactone. The enzyme is highly specific for the B anomer of glucose and does
not affect the a anomer. In spite of this specificity, the reaction catalyzed by glucose oxidase is
commonly used in a clinical assay for total blood glucose -that is, for solutions consisting of a mixture of
B- and a-D-glucose, as well as other sugars present in blood. The oxidation proceeds in the presence of
oxygen and forms hydrogen peroxide, in addition to the lactone. A second enzyme, called peroxidase,
catalyzes the reaction of hydrogen peroxide with colorless compounds to create a colored product,
which is quantified with a simple spectrophotometer. What are the circumstances required to make this
possible? Aside from allowing the detection of smaller quantities of glucose, what advantage does
glucose oxidase offer over the Fehling's reagent for measuring blood glucose?
Chapter 16 Solutions
Concepts of Genetics (11th Edition)
Ch. 16 - Even though the lac Z, Y, and A structural genes...Ch. 16 - Predict the level of genetic activity of the lac...Ch. 16 - Prob. 1CSCh. 16 - Prob. 2CSCh. 16 - Prob. 3CSCh. 16 - Prob. 4CSCh. 16 - HOW DO WE KNOW? In this chapter, we focused on the...Ch. 16 - Prob. 2PDQCh. 16 - Contrast positive versus negative control of gene...Ch. 16 - Contrast the role of the repressor in an inducible...
Ch. 16 - For the lac genotypes shown in the following...Ch. 16 - For the genotypes and conditions (lactose present...Ch. 16 - The locations of numerous lacI and lacIS mutations...Ch. 16 - Prob. 8PDQCh. 16 - Prob. 9PDQCh. 16 - Predict the effect on the inducibility of the lac...Ch. 16 - Erythritol, a natural sugar abundant in fruits and...Ch. 16 - Prob. 12PDQCh. 16 - Prob. 13PDQCh. 16 - Neelaredoxin is a 15-kDa protein that is a gene...Ch. 16 - The creation of milk products such as cheeses and...Ch. 16 - Prob. 16PDQCh. 16 - Prob. 17PDQCh. 16 - Prob. 18PDQCh. 16 - Prob. 19ESPCh. 16 - In a theoretical operon, genes A, B, C, and D...Ch. 16 - A bacterial operon is responsible for the...Ch. 16 - A marine bacterium is isolated and shown to...Ch. 16 - Prob. 23ESPCh. 16 - Prob. 24ESPCh. 16 - One of the most prevalent sexually transmitted...Ch. 16 - Prob. 27ESP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- The M and H subunits of lactate dehydrogenase have very similar sizes and shapes but differ in amino acidcomposition. If the only difference between the two were that theH subunit had a glutamic acid in a position where the M subunithad a serine, how would the five isozymes of LDH separate on electrophoresis using a gel at pH 8.6?arrow_forwardCompare and contrast lactose intolerance with galactosemia.(Hint: Make a table.)arrow_forwardWhat is/are the possible genotype(s) of an individual who is lactose intolerant?arrow_forward
- Lactose permease, a protein of E. coli, is composed of a singlepolypeptide that is 417 amino acids in length. By convention, theamino acids within a polypeptide are numbered from the aminoterminalend to the carboxyl-terminal end. Are the following questionsabout lactose permease true or false?A. Because the sixty-fourth amino acid is glycine and the sixty- eighth amino acid is aspartic acid, the codon for glycine,64, is closer to the 3′ end of the mRNA than the codon for aspartic acid, 68.B. The mRNA that encodes lactose permease must be greater than1241 nucleotides in length.arrow_forwardFailure of anti-oxidant function results in the hydroxylation of an aromatic acid of Enzyme Z and its activation, so that it degrades protoporphyrin to porphyrin, an unstable product. When hit by light, this product further degrades to form a compound responsible for the lesions and excruciating pain the man suffers. The mutation also affected an amino acid at the N-terminal of Enzyme X. Sequencing of the first seven (7) amino acids at the N-terminal of the normal enzyme gave the following sequence: trp-arg-asp-leu-ser-gly-his When the cDNA was sequenced by the Sanger method utilizing ddCTP, the following products were obtained: Tetranucleotide Hexanucleotide Nonanucleotide Decanucleotide Dodenucleotide Octadecanucleotide Nonadecanucleotide 21-nucleotide The mutation involved the 19th bases of the template strand of the peptide. A comparison of the electrophoretic profile of the normal peptide (N) and mutant peptide (M) is shown below. The (+) electrode is situated at the bottom. pH…arrow_forwardFailure of anti-oxidant function results in the hydroxylation of an aromatic acid of Enzyme Z and its activation, so that it degrades protoporphyrin to porphyrin, an unstable product. When hit by light, this product further degrades to form a compound responsible for the lesions and excruciating pain the man suffers. The mutation also affected an amino acid at the N-terminal of Enzyme X. Sequencing of the first seven (7) amino acids at the N-terminal of the normal enzyme gave the following sequence: trp-arg-asp-leu-ser-gly-his When the cDNA was sequenced by the Sanger method utilizing ddCTP, the following products were obtained: Tetranucleotide Hexanucleotide Nonanucleotide Decanucleotide Dodenucleotide Octadecanucleotide Nonadecanucleotide 21-nucleotide What is the sequence of the bases in the mRNA coding for the peptide above?arrow_forward
- Failure of anti-oxidant function results in the hydroxylation of an aromatic acid of Enzyme Z and its activation, so that it degrades protoporphyrin to porphyrin, an unstable product. When hit by light, this product further degrades to form a compound responsible for the lesions and excruciating pain the man suffers. The mutation also affected an amino acid at the N-terminal of Enzyme X. Sequencing of the first seven (7) amino acids at the N-terminal of the normal enzyme gave the following sequence: trp-arg-asp-leu-ser-gly-his When the CDNA was sequenced by the Sanger method utilizing ddCTP, the following products were obtained: Tetranucleotide, Hexanucleotide, Nonanucleotide, Decanucleotide, Dodenucleotide, Octadecanucleotide, Nonadecanucleotide, 21-nucleotide What is the sequence of the bases in the mRNA coding for the peptide above? The mutation involved the 19 bases of the template strand of the peptide. A comparison of the electrophoretic profile of the normal peptided (N) and and…arrow_forwardProteinase K should be used as the first step of DNA purification (before applying phenol CIA mixture) to remove most proteins. Proteinase K alone cannot remove all proteins from the lysate. Explain why the Proteinase K enzyme cannot remove ALL proteins from the cell lysate.arrow_forwardIn biochemistry laboratories, phenylmethylsulfonyl fluoride (PMSF) is a serine protease inhibitor commonly added to cell homogenates to protect proteins from digestion form serine proteases that are released when cell membranes and cell organelles are broken. =S-F PMSF: phenylmethylsulfonyl fluoride 1. Based on our discussion of covalent inhibition of acetylcoline, esterase by diisopropylfluorophosphate, predict the effect of PMSF on chymotrypsin and predict how PMSF will modify the chymotrypsin active site. Justify your answer based on the propertied of the active sites of the enzymes.arrow_forward
- Tazobactam is a BETA-lactam that inhibits the enzyme that bacteria secrete to resist penicillin. Draw a mechanism for the formation of the tazobactam-trapped BETA-lactamase conjugate that proceeds through the following steps.arrow_forwardDescribe type I, II, and III L-asparaginases. Mention characteristics and differences between them with respect to enzyme kinetics (rate, km, etc).arrow_forwardA mutation has occurred that results in phosphofructokinase not being able to bind ATP in its allosteric site. What impact will this mutation have on the production of ATP in the cell? Select one: a. If no ATP can bind to the allosteric site, then phosphofructokinase will not be able to add the phosphate to fructose-6-phosphate to make fructose-1,6-bisphosphate and glycolysis will not work. So no pyruvate, not cellular respiration. b. If ATP cannot bind to the allosteric site, phosphofructokinase will not be activated to make more ATP by substrate-level phosphorylation. c. No impact on the production but will not be able to effectively shut off over production of ATP with feedback inhibition.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Mitochondrial mutations; Author: Useful Genetics;https://www.youtube.com/watch?v=GvgXe-3RJeU;License: CC-BY