
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16.1, Problem 1P
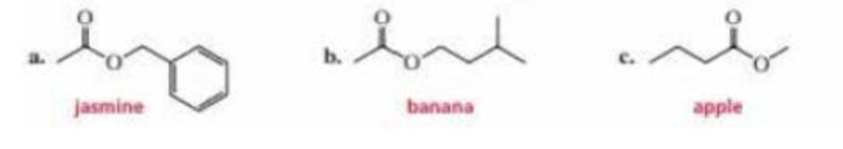
The aromas of many flowers and fruits are due to esters such as those shown in this problem. What are the common names of these esters? (Also see problem 57.)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Question 1
This reaction is an example of a(n) ___________.
E1 reaction
Ester synthesis
Sn2 reaction
pinacol rearrangement
Draw the starting reactant that would produce this hydrazone when treated with this reagent. Ignore inorganic byproducts.
NH2OH, TsOH
What is the importance of proton transfer in the formation of ester?
A.The carbonyl carbon is easily attacked by the nucleophile.
B. The oxygen that has been attached to the two alkyl groups will become stable.
C. The removal of a bulky substituent attached to the reactive site stabilizes the compound.
D. none of the above
Chapter 16 Solutions
Organic Chemistry
Ch. 16.1 - The aromas of many flowers and fruits are due to...Ch. 16.1 - Prob. 2PCh. 16.1 - Name the following:Ch. 16.1 - Prob. 4PCh. 16.2 - Prob. 5PCh. 16.2 - Which is longer, the carbon-oxygen single bond in...Ch. 16.2 - There are three carbon-oxygen bonds in methyl...Ch. 16.2 - Prob. 8PCh. 16.4 - Prob. 9PCh. 16.4 - Prob. 10P
Ch. 16.5 - a. What is the product of the reaction of acetyl...Ch. 16.6 - a. Which compound has the stretching vibration for...Ch. 16.6 - Using the pKa values listed in Table 15.1, predict...Ch. 16.6 - Is the following statement true or false? If the...Ch. 16.7 - What is the product of an acyl substitution...Ch. 16.8 - PROBLEM 16
Starting with acetyl chloride, what...Ch. 16.8 - Prob. 18PCh. 16.9 - We saw that it is necessary to use excess amine in...Ch. 16.9 - Prob. 20PCh. 16.9 - a. state three factors that cause the uncatalyzed...Ch. 16.10 - Prob. 23PCh. 16.10 - Using the mechanism for the acid-catalyzed...Ch. 16.10 - Prob. 25PCh. 16.10 - Prob. 27PCh. 16.10 - Write the mechanism for the acid-catalyzed...Ch. 16.10 - Write the mechanism for the acid-catalyzed...Ch. 16.11 - Prob. 30PCh. 16.12 - Prob. 31PCh. 16.12 - Prob. 32PCh. 16.13 - Prob. 34PCh. 16.13 - Which has a higher melting point, glyceryl...Ch. 16.13 - Draw the structure of an optically inactive fat...Ch. 16.13 - Draw the structure of an optically active fat...Ch. 16.14 - Show how each of the following esters could he...Ch. 16.14 - Prob. 39PCh. 16.15 - Prob. 40PCh. 16.15 - Which of the following reactions leads to the...Ch. 16.16 - Prob. 42PCh. 16.16 - Prob. 43PCh. 16.18 - Prob. 44PCh. 16.18 - Prob. 45PCh. 16.19 - Prob. 46PCh. 16.19 - Which alkyl halides from the carboxylic acids...Ch. 16.20 - Prob. 49PCh. 16.20 - Prob. 50PCh. 16.20 - Prob. 51PCh. 16.21 - Prob. 52PCh. 16.21 - Prob. 53PCh. 16.22 - How could you synthesize the following compounds...Ch. 16 - Prob. 55PCh. 16 - Name the following:Ch. 16 - Prob. 57PCh. 16 - What compound are obtained from the fallowing...Ch. 16 - a. Rank the following esters in order of...Ch. 16 - Because bromocyclohexane is a secondary alkyl...Ch. 16 - a. Which compound would you expect to have a...Ch. 16 - How could you use 1H NMR spectroscopy to...Ch. 16 - Prob. 63PCh. 16 - a. When a carboxylic acid is dissolved in...Ch. 16 - Rank the following compounds in order of...Ch. 16 - Prob. 66PCh. 16 - Prob. 67PCh. 16 - Prob. 68PCh. 16 - A compound with molecular formula C5H10O2 gives...Ch. 16 - Prob. 70PCh. 16 - Prob. 71PCh. 16 - Prob. 72PCh. 16 - Two products, A and B, are obtained from the...Ch. 16 - Prob. 74PCh. 16 - Prob. 75PCh. 16 - Prob. 76PCh. 16 - Prob. 77PCh. 16 - When treated with an equivalent of methanol,...Ch. 16 - a. Identify the two products obtained from the...Ch. 16 - Prob. 80PCh. 16 - Identity the major and minor products of the...Ch. 16 - Prob. 82PCh. 16 - When a compound with molecular formula C11H14O2...Ch. 16 - Prob. 84PCh. 16 - Prob. 85PCh. 16 - The 1H NMR spectra for two esters with molecular...Ch. 16 - Show how the following compounds could be prepared...Ch. 16 - Prob. 88PCh. 16 - Prob. 89PCh. 16 - Prob. 90PCh. 16 - The intermediate shown here is formed during the...Ch. 16 - Prob. 92PCh. 16 - Propose a mechanism that accounts for the...Ch. 16 - Catalytic antibodies catalyze a reaction by...Ch. 16 - Prob. 95P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Please give the major product of the following reactions, this is about amines.arrow_forwardWhich of those would be formed? Then If the product of the reaction in the previous problem was subsequently mixed with sodium cyanoborohydride, which of the following would result? a. primary alcoholb. secondary alcohol c. primary amined. secondary amine e. tertiary aminearrow_forwardUse curved arrows to illustrate how the following decarboxylation occurs in the presence of an acid HA. This reaction constitutes one step in the biosynthesis of the amino acid tyrosine.arrow_forward
- Complete A-> H for this reaction by fill in the blank with appropriate chemicalsarrow_forwardWhat carboxylic acid is used to prepare this ester?arrow_forwardIn Chapter 18, we will learn about the hydrolysis of acetals to aldehydes and ketones. Four of the seven steps in the mechanism for this process are shown in the conversion of acetal A to hemiacetal E. a.Add curved arrows for each step. b.Draw another resonance structure for C. c.Identify the nucleophile and electrophile in Step [3]. d.Which steps are Brønsted–Lowry acid–base reactions?arrow_forward
- Which of the following compounds will form a yellow solid when dissolved in a basic, aqueous solution of I2?(a) butanoic acid; (b) pentan-2-one; (c) pentan-3-one; (d) cyclohexanone; (e) pentanalarrow_forwardWhich functional group has the maximum reactivity toward nucleophilic attack? a) acid chloride b) ketone c) ester d) amidearrow_forwardStarting with benzene, what is a possible product of this series of reactions?: 1) HNO3 & H2SO4 2) Fe, HCl 3) (CH3)2CH-Br, FeBr3 4) NaNO2, HCl, 0-10 oC 5) HBF4 2-fluoro-3-isopropylaniline m-fluoro-benzenesulfonic acid o-fluoro-isopropylbenzene 2-amino-benzenesulfonic acidarrow_forward
- When a molecule has two esters, it can react via an intramolecular Claisen condensation. This is called a Dieckmann condensation. what would be the products of the following reaction (hint: what is going to happen to the protecting group in dilute aqueous acid?).arrow_forwardtrue or false 1. Aldehydes can undergo tautomerization reactions 2. Oxidation reactions are important in excreting carbonyl, nitro and azo groups. 3. First pass effect is a phenomenon in which the drug exhibits its maximum bioavailability 4. Hydrophobic drugs do not undergo significant metabolism 5. In the presence of NADH or NADPH, carbonyl derivatives can be reduced to corresponding alcoholsarrow_forward9. Would thioesters be expected to be more or less reactive to hydrolysis than esters? Explain.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT


Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY