
(a)
Interpretation:
To identify the acyl chloride and amine to form the following amide molecules.
(a) N-ethylbutanamide
Concept introduction:
The preparation amides is done by using carboxylic acids, acyl chlorides and esters. When these carboxylic acid derivatives are treated with ammonia or
The general reaction of acetyl chlorides and amines to synthesize an amide is written as,
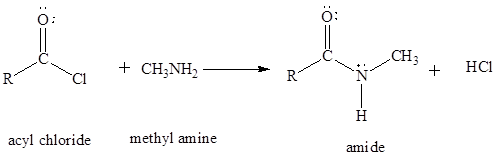
(b)
Interpretation:
To identify the acyl chloride and amine to form the following amide molecules.
(b) N,N-dimethylbenzamide.
Concept introduction:
The preparation amides is done by using carboxylic acids, acyl chlorides and esters. When these carboxylic acid derivatives are treated with ammonia or amines a condensation reaction takes place with the formation of an amide as a major product.
The general reaction of acetyl chlorides and amines to synthesize an amide is written as,
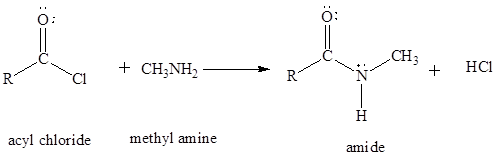
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- Acetaminophen is an analgesic marketed under the brand name Tylenol, among others. Draw the amine that results from the base hydrolysis of acetaminophen.arrow_forwardDraw a structural formula for each amine and amine derivative. (a) N,N-Dimethylaniline (b) Triethylamine (c) tert-Butylamine (d) 1,4-Benzenediamine (e) 4-Aminobutanoic acid (f) (R)-2-Butanamine (g) Benzylamine (h) trans-2-Aminocyclohexanol (i) 1-Phenyl-2-propanamine (amphetamine) (j) Lithium diisopropylamide (LDA) (k) Benzyltrimethylammonium hydroxide (Triton B)arrow_forwardWhich type of amine is phentermine? a) a primary aliphatic amine b) a primary aromatic amine c) a tertiary aliphatic amine d) a tertiary aromatic aminearrow_forward
- Reactions of aldehydes and ketones with amines and amine derivatives a. Draw reaction with a primary amine forms an imine. Hydrazine and hydroxylamine can also be used; they form a hydrazone and an oxime, respectively. b. Draw reaction with a secondary amine forms an enamine.arrow_forwardAromatic amines are _________ neutral because of the polarity are unique as they do not behave like any of the other amines acidic, but stronger than the aliphatic amines basic, but weaker than aliphatic aminesarrow_forwardWhat acyl chloride and amine are required to synthesize the following amides?a. N-ethylbutanamide b. N,N-dimethylbenzamidearrow_forward
- Draw the structure of the product that will be formed when each of the following amines reacts with sodium nitrite and hydrochloric acid, followed by cuprous chloride. (a) propylamine (b) dipropylamine (c) N-propylaniline (d) N,N-dipropylaniline (e) p-propylanilinearrow_forwardFeatures of the structure of imino acids. Formation of amides from amino acids.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole





