
Concept explainers
(a)
Interpretation: The name of the given amine has to be determined.
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
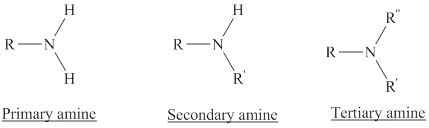
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
(b)
Interpretation: The name of the given amine has to be determined.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
(c)
Interpretation: The name of the given amine has to be determined.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
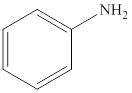
Aniline is an
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Pearson eText Fundamentals of General, Organic, and Biological Chemistry -- Instant Access (Pearson+)
- This is remdesivir, one of the drugs being used in the treatment of COVID-19. Label the indicated carbons as R, S, or neither. b); NH2 HO OHarrow_forwarda) Write out the reaction that converts succinate to fumarate with FAD using curved arrow symbolism. (b) Classify the reaction as an oxidation, reduction, or decarboxylation.arrow_forwardCarvone exists as a pair of enantiomers. (R)-(-)-carvone smells like spearment, whereas (S)-(+)-carvone smells like caraway. Why do these enantiomers have different smells (i.e., different biological activity) ?arrow_forward
- In the monosaccharide derivatives known as sugar alcohols, the carbonyl oxygen is reduced to a hydroxyl group. For example, D-glyceraldehyde can be reduced to glycerol. However, this sugar alcohol is no longer designated D or L. Why?arrow_forwardDraw all possible carboxylic acids with the formula C5H10O2.arrow_forwardHow many trimethylcyclopentanes are possible?arrow_forward
- name the oxidation/reduction product for the given monosaccharidearrow_forwardWhat is the ionisable group of phenazopyridine? With illustrationsarrow_forwardD- and L- designations are used to distinguish between the two possible enantiomers of the monosaccharide, galactose. From the Fischer projection, determine the designation of this monosaccharide.arrow_forward
- Change the structure in the drawing area to show the aldonic acid product formed if the compound is easily oxidized. If there are no changes to be made, check the No changes box under the drawing area. No changes × CH₂OH C: H -OH H -OH H -OH снонarrow_forwardFollowing are Fischer projections for a groupof five-carbon sugars, all of which are aldopentoses. Identify thepairs that are enantiomers and the pairs that are epimers. (Thesugars shown here are not all of the possible five-carbon sugars.)arrow_forwardDraw the structures of the following compounds. (Includes both new and old names.) 3-cyclopentylhexan-3-olarrow_forward
