
Concept explainers
(a)
Interpretation:
The organic reactant in the given reaction equation has to be named.
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
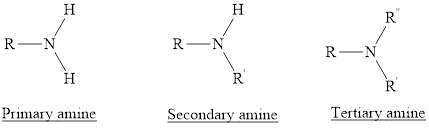
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Amines form salts by accepting a proton from strong mineral acids.
(b)
Interpretation:
The organic reactant in the given reaction equation has to be named
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Reaction of amines and acid will give amine salt an (ammonium ion).
Treating an amine salt with a strong base regenerates the “parent” amine.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
(c)
Interpretation:
The organic reactant in the given reaction equation has to be named
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Amines form salts by accepting a proton from strong mineral acids.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Pearson eText Fundamentals of General, Organic, and Biological Chemistry -- Instant Access (Pearson+)
- Change the structure in the drawing area to show the aldonic acid product formed if the compound is easily oxidized. If there are no changes to be made, check the No changes box under the drawing area. No changes × CH₂OH C: H -OH H -OH H -OH снонarrow_forwardThe pOH of a basic solution is 3.75. What is [H⁺]?arrow_forwardSalicylic acid (o-hydroxybenzoic acid) is used as starting material to prepare aspirin. Draw the structure of salicylic acid.arrow_forward
- 49.10 Lactic Acid Salicylic Acid 48 5 g 2g Trichloroacetic Acid Flexible Collodion q.s. ad 100 g Sig: wart remover. Use as directed. (a) Flexible collodion contains 20% w/w camphor and 30% w/w castor oil. How many grams of each would be contained in 30 g of the mixture? (b) The specific gravity of castor oil is 0.955. How many milliliters of the oil are contained in 30 g of the mixture? (c) If the specific gravity of the mix- ture is 0.781, what are the w/v con- centrations of lactic acid, salicylic acid, and trichloroacetic acid in the mixture? 49. (a) 5.34 g camphor and 8.01 g castor oil (b) 8.39 mL castor oil (c) 3.12% w/v lactic acid, 3.91% w/v sali- cylic acid, and 1.56% w/v trichloroacetic acidarrow_forwardJust question 7.52arrow_forwardThe chemical formula for deoxyribose is C___ H___O____.arrow_forward
- If you need to make 2L of 0.25M Glucose, how many grams of glucose do you need Formula for glucose: C6H12O6arrow_forward75 mL of 0.300 mol/L sodium phosphate solution is combined with 67.5 mL of 0.350 mol/L calcium bicarbonate. a)Before you begin your reaction, you must accurately produce 1.500 L of your sodium phosphate solution from sodium phosphate trihydrate solid. Write out a procedure to explain all the steps you will take in the lab when making the solution to ensure that your solution concentration is accurate. Please include calculations that show the required mass of solid. Also include the correct names of all equipment used. b)You have a super powerful microscope in your lab! You are able to zoom in on your sodium phosphate solution and take a picture at the molecular level. Label the diagram on the left with the correct choices from the box on the right. You may use arrows or rewrite the symbols in one appropriate place. c)In one sentence, explain what the diagram is showing.arrow_forwardColumn A shows the names of some of the c functional groups. Column B shows their structure. Match each entry in column A one in column B.arrow_forward
 Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College
Anatomy & PhysiologyBiologyISBN:9781938168130Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark WomblePublisher:OpenStax College

