
Concept explainers
(a)
Interpretation: The structure of the given amine has to be determined.
Concept introduction:
In chemistry Structure is the arrangement of
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
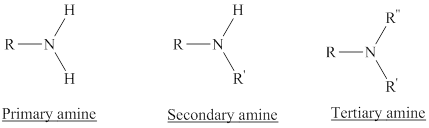
From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
(b)
Interpretation: The structure of the given amine has to be determined.
Concept introduction:
In chemistry Structure is the arrangement of chemical bonds between atoms in a molecule, specifically which atoms are chemically bonded to what other atoms with what kind of chemical bond.
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
(c)
Interpretation: The structure of the given amine has to be determined.
Concept introduction:
In chemistry Structure is the arrangement of chemical bonds between atoms in a molecule, specifically which atoms are chemically bonded to what other atoms with what kind of chemical bond.
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Aniline is an
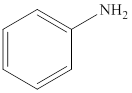
(d)
Interpretation: The structure of the given amine has to be determined.
Concept introduction:
In chemistry Structure is the arrangement of chemical bonds between atoms in a molecule, specifically which atoms are chemically bonded to what other atoms with what kind of chemical bond.
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.

From the name of the compound its structure can be determined.
Primary amines can be named in the IUPAC system in several ways,
For simple amines the suffix – amine is added to the name of the alkyl substituent.
The suffix-amine can be used in place of the final –e in the name of the parent compound.
For a secondary amine an N prefixes the compound giving the shorter carbon chain and its chain prefix name.
For a tertiary amine an N, N prefixes the compound giving the two shorter carbon chains and their side chain prefix names.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Pearson eText Fundamentals of General, Organic, and Biological Chemistry -- Instant Access (Pearson+)
- Draw the structures of the following acids:(a) 2-Ethyl-3-hydroxyhexanoic acid (b) m-Nitrobenzoic acidarrow_forwardDraw a structural formula for each compound. (a) trans-2-Methyl-3-hexene(b) 2-Methyl-3-hexyne(c) 2-Methyl-1-butene (d) 3-Ethyl-3-methyl-1-pentyne (e) 2,3-Dimethyl-2-pentenearrow_forward(A) What are Waxes? Draw the structure of wax, which is made up of palmitic acid (16:0) and a saturated 18-carbon alcohol. (В) Consider the structure of menthol. How many isoprene units are present in menthol? (C) What type of isoprene linkage (head-to-tail or tail-to-tail) is present in menthol? Identify the isoprene linkage in the following structure of menthol and indicate by a circle. Menthol =arrow_forward
- Which of the following define the stereochemistry of alanine (as per the structure shown)? Note: Functional groups arranged horizontally are facing towards the front, and the functional groups arranged vertically are facing towards the back. a) d- b) S- c) R- d) I-arrow_forwardA) Describe the glycosidic bond (using standard convention) indicated by “Arrow a.” B) Draw the open chain Fischer projection formula of the monosaccharide labeled “B” C) Describe the glycosidic bond (as in question A) indicated by “Arrow b.”arrow_forwardDraw the structures of the following compounds. (Includes both new and old names.) 3-cyclopentylhexan-3-olarrow_forward
- Are the following molecules chiral or achiral? If they are chiral, identify the chiral carbon atom(s).(a) Pentan-3-ol (b) 2-Bromobutane(c) 2-Methylcyclohexanolarrow_forwardSpermaceti, a fragrant substance isolated from sperm whales, was commonly used in cosmetics until it was banned in 1976 to protect the whales from extinction. Chemically, spermaceti is cetyl palmitate, the ester of palmiticacid with cetyl alcohol (the straight-chain 16-carbon alcohol). Draw the structure of spermaceti.arrow_forwardIdentify the acid on the left and its conjugate base on the right in the following equations:(a) HOCl + H2O ↔ H3O+ + OCl-(b) HONH2 + H2O ↔ HONH3+ + OH-(c) NH4+ + H2O ↔ NH3 + H3O+(d) 2HCO3-2 ↔ H2CO3 + CO3-2 (e) PO4-3 + H2PO4- ↔ 2HPO4-2arrow_forward
- Indicate the following for each disaccharide: I) type of glycosidic linkage i.e. a(1→4) II) configuration i.e. alpha or beta 1. OH CH₂OH 0 OH I) II) III) IV) OH CH₂OH -0 OH ОН OH 2. HO CH₂OH 0 I) II) III) IV) ОН HOCH2 ОН HO ОН CH₂OH 3. CH, OH CH2 О CG OH ОН HO ОН HỎ I) II) III) III) reducing or non-reducing sugar IV) name of each monosaccharide IV) OH OH 4. HO I) II) III) IV) CH₂OH OH 0 ОН CH₂OH OH ОН OHarrow_forward2) Draw the skeletal (line-boad) structure for 2-methylbutanediaicacid. 3) Draw the skeletal structuce for (E)-3-phenylbut - 2-cnois acid. 4) Draw the skele tal structure for (2)-3-chloro-2- phenylprop-2-enaic acid. 5) The common fea ture of all esters is the presence of a A)-OH group B)-CE8)OH group C)-C(=0)H group D)-C(=0)OR group. 6) Draw the skeletal structuce of 3,3-dimethylbutanenitrilearrow_forwardA pentapeptide on complete hydrolysis yields 3 moles of glycine (R=H), 1 mole of alanine (R=CH3) and 1 mole of serine (R=CH2OH). On partial hydrolysis, the fragments are ala-gly, gly-ala and gly-ser. The pentapeptide is negative to the Sanger reaction. Draw a possible structure for the pentapeptide.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





