
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
4th Edition
ISBN: 9780134465715
Author: John E. McMurry, David S. Ballantine
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 16.2, Problem 16.6KCP
Interpretation Introduction
Interpretation: The condensed and line structure formula of the given compound has to be drawn. It should be find out whether the compound is primary, secondary or tertiary.
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
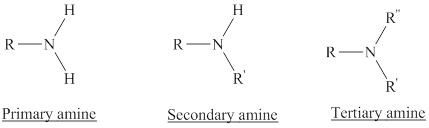
In bond Line structure,
- Only shows bonds.
- C atoms assumed at each end and intersection of bonds
- H atoms are not shown.
- Assume 4 bonds to each C
- Fulfill C’s 4 bonds by adding hydrogens.
In condensed structure representation,
- Some or all of the lines are omitted and atoms attached to carbon are written immediately after it.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For A, B, C, D, E, F, identify the circled functional groups and linkages in the compound in the picture.
With the given structures, arrange the following compounds in order of decreasing reactivity towards hydrolysis
What prefixes are used in naming the following?(a) A 1,3-disubstituted benzene(b) A 1,4-disubstituted benzene
Chapter 16 Solutions
Fundamentals of General, Organic, and Biological Chemistry, Books a la Carte Edition; Modified Mastering Chemistry with Pearson eText -- ValuePack ... and Biological Chemistry (4th Edition)
Ch. 16.2 - Identify the following compounds as primary,...Ch. 16.2 - Prob. 16.2PCh. 16.2 - Prob. 16.3PCh. 16.2 - Prob. 16.4PCh. 16.2 - Prob. 16.5KCPCh. 16.2 - Prob. 16.6KCPCh. 16.3 - Arrange the following compounds in order of...Ch. 16.3 - Draw the structures of (a) ethylamine and (b)...Ch. 16.4 - Provide compounds that fit the following...Ch. 16.4 - Prob. 16.10P
Ch. 16.4 - Prob. 16.11PCh. 16.5 - Write an equation for the acid-base equilibrium...Ch. 16.5 - Prob. 16.13PCh. 16.5 - Prob. 16.14PCh. 16.5 - Prob. 16.15PCh. 16.5 - Prob. 16.16PCh. 16.6 - Prob. 16.17PCh. 16.6 - Prob. 16.18PCh. 16.6 - Prob. 16.19PCh. 16.6 - Prob. 16.20PCh. 16.6 - Prob. 16.21PCh. 16.6 - Prob. 16.22PCh. 16.7 - Prob. 16.1CIAPCh. 16.7 - Prob. 16.2CIAPCh. 16.7 - Prob. 16.3CIAPCh. 16 - (a) For the compound above, identify each nitrogen...Ch. 16 - The structure of the amino acid lysine (in its...Ch. 16 - Prob. 16.25UKCCh. 16 - Prob. 16.26UKCCh. 16 - Prob. 16.27UKCCh. 16 - Complete the following equations: (a) (b)...Ch. 16 - Prob. 16.29APCh. 16 - Draw the structures corresponding to the following...Ch. 16 - Name the following amines, and classify them as...Ch. 16 - Name the following amines, and identify them as...Ch. 16 - Prob. 16.33APCh. 16 - Which is a stronger base, diethyl ether or...Ch. 16 - Prob. 16.35APCh. 16 - Prob. 16.36APCh. 16 - The compound lidocaine is used medically as a...Ch. 16 - Prob. 16.38APCh. 16 - Draw the structures of the ammonium ions formed...Ch. 16 - Prob. 16.40APCh. 16 - Prob. 16.41APCh. 16 - Prob. 16.42APCh. 16 - Prob. 16.43APCh. 16 - Prob. 16.44APCh. 16 - Prob. 16.45CPCh. 16 - Prob. 16.46CPCh. 16 - Prob. 16.47CPCh. 16 - Prob. 16.48CPCh. 16 - How do amines differ from analogous alcohols in...Ch. 16 - Name at least two undesirable characteristics are...Ch. 16 - Prob. 16.52CPCh. 16 - Complete the following equations (Hint: Answers...Ch. 16 - Prob. 16.54CPCh. 16 - Prob. 16.55CPCh. 16 - Why is cyclohexylamine not considered to be a...Ch. 16 - Prob. 16.57CPCh. 16 - Prob. 16.58GPCh. 16 - 1-Propylamine, 1-propanol, acetic acid, and butane...Ch. 16 - Prob. 16.60GPCh. 16 - Lemon juice, which contains citric acid, is...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Write the chemical formula for the conjugate base of each of the following acids: Carbonic Acid (H2CO3)___________________ Formic Acid (CH2O2)_____________________arrow_forwardHow many total isomers are possible in the picture?arrow_forwardIdentify the functional groups in the following compoundsarrow_forward
- draw a fischer projection formula for the enantiomer of each of the following monosaccharides.arrow_forwardWhy is the electronic geometry of water molecule tetrahedral, but we describe the geometry as bent?arrow_forwardConsider the following acids and their ionization constant, determine which conjugate base is HCOOH Ka = 1.7 x 10-4 (b) HCN Ka = 4.9 x 10-10arrow_forward
- Which of the following Fischer formula is or are monosaccharide that has two chiral centers?arrow_forwardwhich of the following functional groups are present in this molecule? amine amide amino acid keytone ether could you also identify this molecule?arrow_forwardWhy is this compound achiral?arrow_forward
- What are the names of the seven functional groups found in organic molecules? Underline the two that can act as an acid. Make the single group that acts as a base in bold lettering.arrow_forwardPlease explain the following terms: Chirality Enantiomer Stereoisomer Diastereomerarrow_forwardWhy are disulfide bonds particularly important and unique in their role as determinants of tertiary structure?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you


Macromolecules | Classes and Functions; Author: 2 Minute Classroom;https://www.youtube.com/watch?v=V5hhrDFo8Vk;License: Standard youtube license