
Concept explainers
(a)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
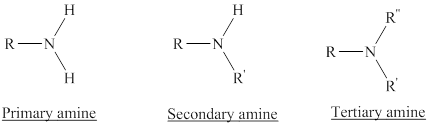
In a quaternary ammonium ion a nitrogen atom with four attached groups is positively charged. And their compounds are known as quaternary ammonium salt.
Reactions of these types of amine with
(b)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
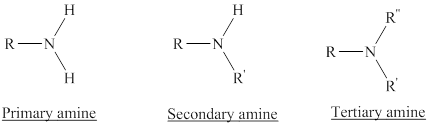
In a quaternary ammonium ion a nitrogen atom with four attached groups is positively charged. And their compounds are known as quaternary ammonium salt.
Reactions of these types of amine with
(c)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
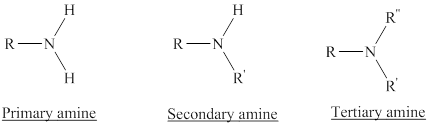
In a quaternary ammonium ion a nitrogen atom with four attached groups is positively charged. And their compounds are known as quaternary ammonium salt.
Reactions of these types of amine with
(d)
Interpretation:
The given reaction equation should be completed.
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
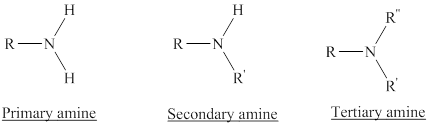
In a quaternary ammonium ion a nitrogen atom with four attached groups is positively charged. And their compounds are known as quaternary ammonium salt.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
- Please only provide the solution of the following as quick as possible:arrow_forwardGive the equation to calculate thoracic fraction (TPM)?arrow_forwardUsing the formula V₁D₁ = V₂D2, what volume of a 1/100 (10-2) dilution would you need to produce 100 mL of a 1/10000 (10-4) dilution? (Note: provide your answer in decimal format to three decimal places.)arrow_forward

