
Concept explainers
Interpretation:
It should be determined that which of the two
Concept introduction:
Amines are the derivatives of ammonia
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
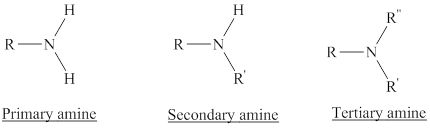
Hydrogen bond is an attractive force established between hydrogen atom attached to a highly electronegative element and another highly electronegative element of the same or different molecule.
High solubility is expected when hydrogen bonds form between solute and solvent molecule.
Solubility will decrease with increase in alkyl groups in a compound.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
- Why would a crystal of NaCl dissolve in water?arrow_forwardWhich of the following group is present in thymine but absent in uracil? NH3 COOH C=O OCH3 CH3arrow_forwardWhat structural similarities exist between a triacylglycerol and a phosphatidyl ethanolamine? How are these two kinds of lipids structurally distinct?arrow_forward
- Given the following information about amino acid tyrosine answer questions 1 & 2: A. O H HO OH HO HO рказ 10.1 H NH₂ CH₂OH A. NH3 С. НО 1. Which of the above forms of tyrosine will be predominant in a solution with pH 9.5? 2. Which of the above forms of tyrosine is a cation? Answers to Questions 3 & 4 should be selected from the following choices: CH₂OH Holl O OH B. NH₂ pka2 9.01 conj acid OH В. НО "OH D. HO HO H OH pka1 2.2 O H -OH CH₂OH C. NH3 HO NH₂ O HO OH D. 3. Which of the monosaccharides above is a non-reducing sugar? 4. Which of the monosaccharides above is an aldotetrose? 5. What is the main functional group in a protein? ||||OHarrow_forwardCatalytic hydrogenation, used in the food industry, converts double bonds in the fatty acids of the oil triacylglycerols to —CH2— CH2—. How does this affect the physical properties of the oils?arrow_forwardWhat is the pH of a glycine solution in which the alpha-carboxyl group is one third dissociated? Given pka1=2.3, pka2=9.6arrow_forward
- Why are some solutes soluble in water and solutes soluble in cyclohexane ?arrow_forwardOne of these forms of cocaine is relatively insoluble inwater: which form, the free base or the hydrochloride?arrow_forwardWhat structural features do a triacylglycerol and a phosphatidyl ethanolamine have in common? How do the structures of these two types of lipids differ?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





