
Concept explainers
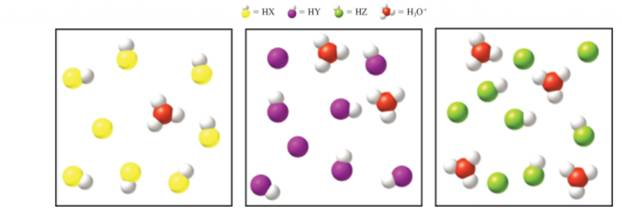
The diagrams show solutions of three different weak acids with formulas
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Looseleaf Study Guide For Chemistry
- Write equations to illustrate the acid-base reaction when each of the following pairs of Brnsted acids and bases are combined: Acid Base a.HOCl H2O b.HClO4 NH3 c.H2O NH2 d.H2O OCl e.HC2O4 H2Oarrow_forwardEach box represents an acid solution at equilibrium. Squares represent H+ ions, and circles represent the anion. Water molecules are not shown. Which figure represents a strong acid? Which figure is a weak acid?arrow_forwardIn each of the following chemical equations, identify the conjugate acid—base pairs. a. HF(aq)+H2O(l)F-(aq)+H3O+(aq)b. CN(aq)+H2O(l)HCN(aq)+OH(aq) c. HCO3-(aq)+H2O(l)H2CO3(aq)+OH-(aq)arrow_forward
- . Using Fig. 16.3, list the approximate pH value of live “everyday” solutions. How do the familiar properties (such as the sour taste for acids) of these solutions correspond to their indicated pH?arrow_forwardUse Table 14.3 to help order the following acids from strongest to weakest HNO3,H2O,NH4+,C5H5NH+arrow_forwardConsider two separate solutions: one containing a weak acid. HA, and one containing HCl. Assume that you start with 10 molecules of each. a. Draw a molecular-level picture of what each solution looks like. b. Arrange the following from strongest to weakest base: Cl- H2O. A- . Explain.arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





