
Concept explainers
(a)
Interpretation:
The principal organic compound that is expected when
Concept introduction:
The addition reaction of the
Answer to Problem 17.22AP
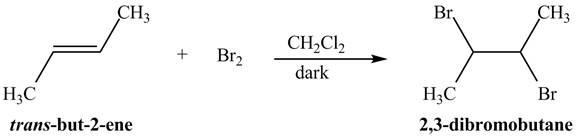
The principal organic compound that is formed when
Explanation of Solution
The reaction of
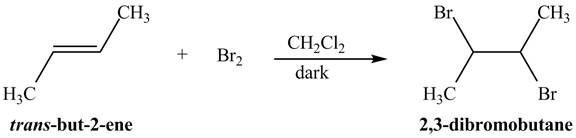
Figure 1
The addition of the bromine molecule on the double bond of the alkene takes place to give the dibromo product. The addition of bromine to the double bond is the anti-addition.
The principal organic compound that is formed when
(b)
Interpretation:
The principal organic compound that is expected when
Concept introduction:
The addition reactions of the alkene are very well known reactions. The electron density on the alkene double bond makes it nucleophilic. The alkene undergoes varieties of addition reaction via different mechanisms.
Answer to Problem 17.22AP
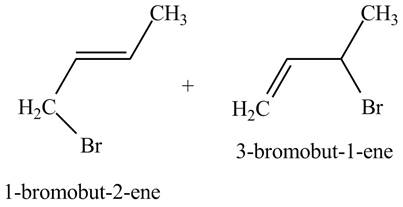
The principal organic compound that is formed when
Explanation of Solution
The reaction of
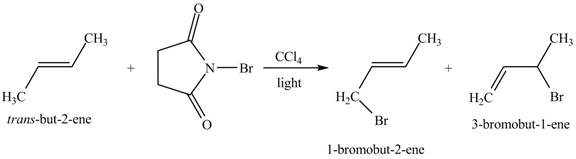
Figure 2
The reaction of an alkene with N-bromosuccinimide in the presence is the free-radical reaction. In this reaction, allylic bromination is observed instead of an addition to the alkene. The N-bromosuccinimide acts as radical initiator. This molecule breaks the
The principal organic compound that is formed when
(c)
Interpretation:
The principal organic compound that is expected when product(s) of part (b) undergo solvolysis in aqueous acetone is to be stated.
Concept introduction:
The nucleophilic substitution reactions are the reactions in which a group is substituted by a nucleophile. The
The
Answer to Problem 17.22AP
The principal organic compounds that are formed when product(s) of part (b) undergo solvolysis in aqueous acetone are shown below.
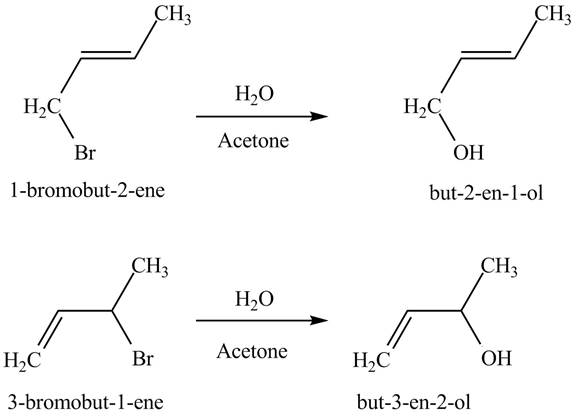
Explanation of Solution
The products of part (b) are shown below.
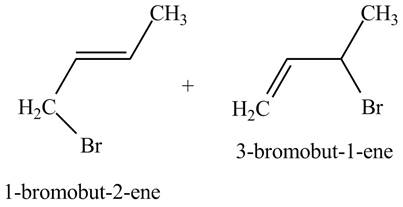
Figure 3
The reaction when product(s) of part (b) undergo solvolysis in aqueous acetone is shown below.
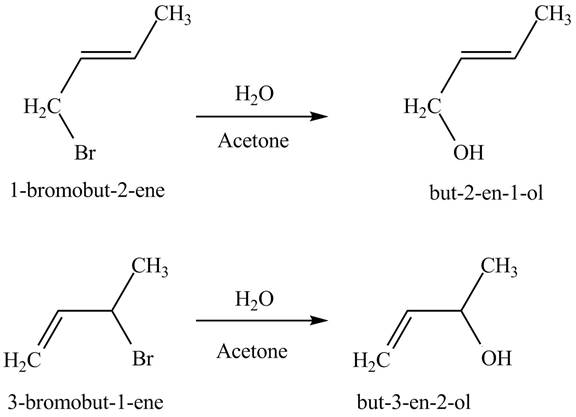
Figure 4
The nucleophilic substitution reaction takes place when solvolysis of products of part (b) takes place. The bromine group is substituted by the hydroxyl group giving rise to the formation of an allylic alcohol.
The principal organic compound that is formed when product(s) of part (b) undergo solvolysis in aqueous acetone is shown in Figure 4.
(d)
Interpretation:
The principal organic compound that is expected when product(s) of part (b) are reacted with
Concept introduction:
The reaction of an
Answer to Problem 17.22AP
The principal organic compounds that are formed when product(s) of part (b) are reacted with
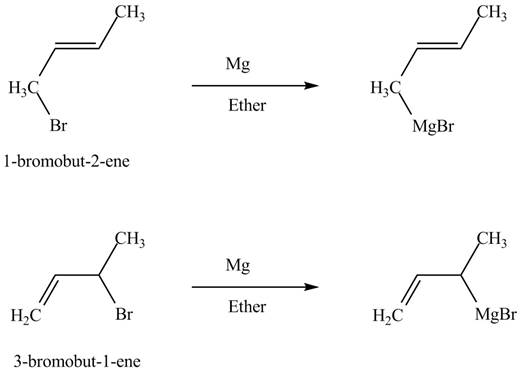
Explanation of Solution
The products of part (b) are shown below.
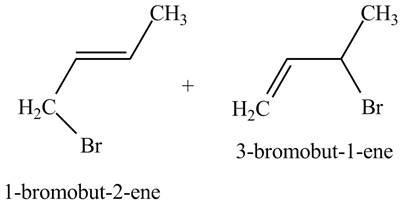
Figure 3
The reaction that occurs when product(s) of part (b) are reacted with
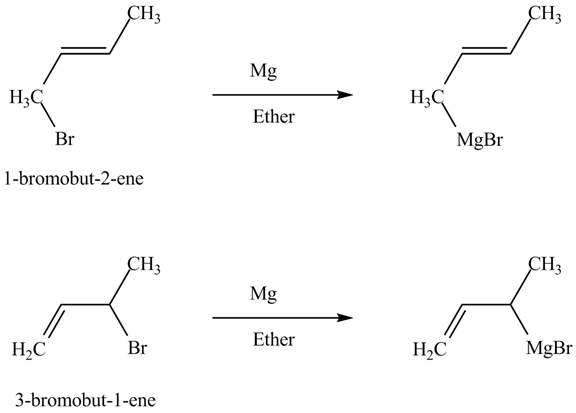
Figure 5
The allylic halide also undergoes the same kind of reactions are do the alkyl halide with magnesium metal in dry ether. They are also lead to the formation of Grignard reagent but this time with the allylic group.
The principal organic compound that is formed when product(s) of part (b) undergo solvolysis in aqueous acetone is shown in Figure 5.
(e)
Interpretation:
The principal organic compound that is expected when product(s) of part (d) are reacted with
Concept introduction:
The reaction of an alkyl halide with a metal-like magnesium in the presence of dry ether leads to the formation of
Answer to Problem 17.22AP
The principal organic compound that is formed when products of part (d) react with
Explanation of Solution
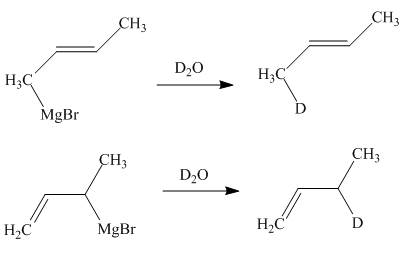
The products of part (d) are shown below.
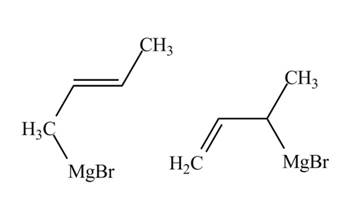
Figure 6
The Gringnard reagents react with

Figure 7
The principal organic compound that is formed when products of part (d) react with
Want to see more full solutions like this?
Chapter 17 Solutions
ORGANIC CHEMISTRY SAPLING ACCESS + ETEX
- Mention and Write the following reaction mechanisms: a) thermal dehydration of 4-t-butylcyclohexanol with phosphoric acid as catalyst; b) addition of molecular bromine to the C= C double bond of 4-t- butylcyclohexene;arrow_forwardWhen A is reacted with hot aqueous NaOH, a compound B of molecular formula C8H11NO is produced. With this information, write the correct structure of B and propose the reaction mechanism (step by step, with the correct use of arrows) to obtain B.arrow_forwardA certain hydrocarbon, C7H10, yields the two products shown below upon oxidative cleavage with KMnO4/H3O+ . Deduce the structure of the hydrocarbon.arrow_forward
- A. In the synthesis of 1-bromobutane, what is the inorganic by-product left in the reaction flask following the distillation? Why was the bromoalkane the bottom layer in the separatory funnel? B. Predict the product when 1-methylcyclohexanol reacts with H2SO4 and KBr. Show the mechanism.arrow_forwardA chemist needs an ether to use as a solvent for a reaction and wants to synthesize it in one step from two of the following available reagents: sodium ethoxide, bromomethane, potassium tert-butoxide, and 2-bromo-2-methylpropane. i) Which combination(s) will give a good yield of an ether? Illustrate, showing the mechanism of the reaction. ii) Illustrate with a mechanism the reaction of one of the combinations that will not yield an ether?arrow_forwardmost of the additions of bromine to double bonds gave entirely antistereochemistry. Explain why the addition to phenanthrene gives a mixture of synand anti stereochemistry. When the product from (c) is heated, HBr is evolved and 9-bromophenanthrene results.Propose a mechanism for this dehydrohalogenation.arrow_forward
- The following bicyclic ketone has two -carbons and three -hydrogens. When this molecule is treated with D2O in the presence of an acid catalyst, only two of the three -hydrogens exchange with deuterium. The -hydrogen at the bridgehead does not exchange. How do you account for the fact that two -hydrogens do exchange but the third does not? You will find it helpful to build models of the enols by which exchange of -hydrogens occurs.arrow_forwardDeduce the structure of each compound from the information given. All unknowns in this problem have molecularformula C8H12.(a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis of W, followed by reduction with dimethylsulfide, gives octanedioic acid, HOOC¬(CH2)6¬COOH. Draw the structure of W.(b) Upon catalytic hydrogenation, unknown X gives cyclooctane. Ozonolysis of X, followed by reduction with dimethylsulfide, gives two equivalents of butanedial, O“CH¬CH2CH2¬CH“O. Draw the structure of X.(c) Upon catalytic hydrogenation, unknown Y gives cyclooctane. Ozonolysis of Y, followed by reduction with dimethylsulfide, gives a three-carbon dialdehyde and a five-carbon dialdehyde. Draw the structure of Y.*(d) Upon catalytic hydrogenation, unknown Z gives cis-bicyclo[4.2.0]octane. Ozonolysis of Z, followed by reductionwith dimethyl sulfide, gives a cyclobutane with a three-carbon aldehyde (¬CH2¬CH2¬CHO) group on C1 and aone-carbon aldehyde (¬CHO) group on C2. Draw the…arrow_forwarda) Give the appropriate base for the above reaction and draw the resonance structures of the enolate ions derived from compound A. Then write the mechanism for the formation of the Dieckmann cyclized product B. b) If compound B above is reacted with NaBH 4 , draw the structure of the reduced product. Give a reason for your choice of product. c) Being a β-ketoester, B could undergo a 3-step synthesis involving alkylation, hydrolysis and decarboxylation reactions to yield the cyclopentanone, C. Write the outline synthesis for each step which include the appropriate regents and correct intermediate.arrow_forward
- 3. Provide a detailed, stepwise mechanism for the acid-catalyzed reaction of 2-butanone with ethylene glycol (HOCH2CH2OH) to produce an acetal.arrow_forwardAcid-catalyzed dehydration of neopentyl alcohol, (CH3)3CCH2OH, yields 2-methyl-2- butene as the major product. Outline a mechanism showing all steps in its formation.arrow_forwardProvide the reagents necessary to convert 3-methyl-2-butanol to 2-bromo-3-methylbutane. CHOOSE FROM THE FOLLOWING REAGRENTS BELOW conc. HBr NaBr, H2SO4 PBr3 HBr, peroxide Br2arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

