
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
8th Edition
ISBN: 9781305582439
Author: Brown, William H.; Iverson, Brent L.; Anslyn, Eric; Foote, Christopher S.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 17, Problem 17.45P
Interpretation Introduction
Interpretation:
A reaction roadmap has to be made for the reactions in the study Guide section of
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
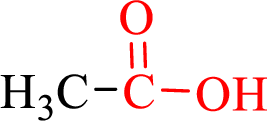
The various sections in the study guide part in the chapte named carbxylic acid are mentioned below,
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
help
20:32
hydroxide
2. Using curved arrows, show the deprotonation mechanism of phenol with
(OH). Also, provide the structure of the product of this deprotonation.
Start answering this question by drawing the structure of phenol. Then
hydroxide ion. Find your most reactive electrons and draw the curved arrow that
shows where they would go to deprotonate phenol. Then "make a bond break a
bond"
write they
question
3. Provide valid resonance structures for the product of
the previous
arrow(s)
charge moving onto the ortho- and para positions. Use curved
to show how the charge resonates to the next structure in your scheme.
all the proper notations for depicting resonance.
that show the
in the structures
Be sure to use
Send a chat
C
Using your reaction roadmap as a guide, show how to convert ethylene into 1-butene.
All of the carbon atoms of the target molecule must be derived from ethylene. Show all
intermediate molecules synthesized along the way.
Ethylene
1-Butene
Chapter 17 Solutions
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
Ch. 17.2 - Prob. 17.1PCh. 17.4 - Which is the stronger acid in each pair?Ch. 17.4 - Prob. 17.3PCh. 17.7 - Prob. 17.4PCh. 17.8 - Prob. 17.5PCh. 17.8 - Prob. AQCh. 17.8 - Prob. BQCh. 17.8 - Prob. CQCh. 17.8 - Permethrin and Bifenthrin Pyrethrin is a natural...Ch. 17.9 - Prob. 17.6P
Ch. 17 - Write the IUPAC name of each compound, showing...Ch. 17 - Prob. 17.8PCh. 17 - Prob. 17.9PCh. 17 - Prob. 17.10PCh. 17 - Prob. 17.11PCh. 17 - Prob. 17.12PCh. 17 - Prob. 17.13PCh. 17 - On a cyclohexane ring, an axial carboxyl group has...Ch. 17 - Prob. 17.15PCh. 17 - Prob. 17.16PCh. 17 - Prob. 17.17PCh. 17 - Complete each reaction.Ch. 17 - Prob. 17.19PCh. 17 - Prob. 17.20PCh. 17 - Prob. 17.21PCh. 17 - Show the reagents and experimental conditions...Ch. 17 - Prob. 17.23PCh. 17 - Prob. 17.24PCh. 17 - Prob. 17.25PCh. 17 - In each set, assign the acid its appropriate pKa.Ch. 17 - Low-molecular-weight dicarboxylic acids normally...Ch. 17 - Complete the following acid-base reactions. (a)...Ch. 17 - Prob. 17.29PCh. 17 - Prob. 17.30PCh. 17 - Excess ascorbic acid is excreted in the urine, the...Ch. 17 - Give the expected organic product when...Ch. 17 - Show how to convert trans-3-phenyl-2-propenoic...Ch. 17 - Show how to convert 3-oxobutanoic acid...Ch. 17 - Prob. 17.35PCh. 17 - Prob. 17.36PCh. 17 - Prob. 17.37PCh. 17 - When 4-hydroxybutanoic acid is treated with an...Ch. 17 - Fischer esterification cannot be used to prepare...Ch. 17 - Draw the product formed on thermal decarboxylation...Ch. 17 - Prob. 17.41PCh. 17 - Show how cyclohexanecarboxylic acid could be...Ch. 17 - Prob. 17.43PCh. 17 - Prob. 17.44PCh. 17 - Prob. 17.45PCh. 17 - Write the products of the following sequences of...Ch. 17 - Using your reaction roadmaps as a guide, show how...Ch. 17 - Using your reaction roadmaps as a guide, show how...Ch. 17 - Using your reaction roadmaps as a guide, show how...Ch. 17 - Using your reaction roadmaps as a guide, show how...Ch. 17 - Prob. 17.51PCh. 17 - Complete the following Fischer esterification...Ch. 17 - Prob. 17.53P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Using your reaction roadmap as a guide, show how to convert butane into butanal. Show all reagents needed and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmaps as a guide, show how to convert propane into propyl propanoate. You must use propane as the source of all carbon atoms in the target molecule. Show all reagents needed and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmap as a guide, show how to convert butane into 2-butyne. Show all reagents and all molecules synthesized along the way. Butane 2-Butynearrow_forward
- 1 Using your reaction roadmap as a guide, show how to convert butane into butanal. Show all reagents needed and all molecules synthesized along the way. Butane Butanalarrow_forwardUsing your reaction roadmap as a guide, show how to convert 1-bromopropane and carbon dioxide into 4-propyl-4-heptanol. You must use 1-bromopropane and carbon dioxide as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmap as a guide, show how to convert cyclohexane into hexanedial. Show all reagents and all molecules synthesized along the way.arrow_forward
- Using your reaction roadmap as a guide, show how to convert propane into butyronitrile. You must use propane and sodium cyanide as the source of all of the carbon atoms in the butyronitrile product. Show all reagents and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmap as a guide, show how to convert 1-propanol into 2-hexyne. You must use 1-propanol as the source of all carbon atoms in the target molecule. Show all reagents needed and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmap as a guide, show how to convert 1-bromopentane and sodium cyanide into N-hexylhexanamide. You must use 1-bromopentane and sodium cyanide as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forward
- Using your reaction roadmaps as a guide, show how to convert ethanol into 2-pentanone. You must use ethanol as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forwardUsing your reaction roadmaps as a guide, show how to convert 4-methyl-1-pentene and carbon dioxide into 5-methylhexanoic acid. You must use 4-methyl-1-pentene and carbon dioxide as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way.arrow_forwardUse a series of reactions to show how you can make 2-pentanone and 3-pentanone from 2-pentyne. Name all reactants and intermediate products formed. Indicate the type(s) of reactions required.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning