
(a)
Interpretation: The equation for the reaction of acid in Example 17.3 with ammonia has to be written and the name of the
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,
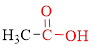
Nomenclature of carboxylic acid:
- Find the Parent hydrocarbon chain.
- Carboxyl carbon must be numbered first.
- Replace the –e in the
alkane name with –oic acid.
Carboxylic acid reacts with ammonia gives neutral carboxylic salt.
(b)
Interpretation: The equation for the reaction of acid in Example 17.3 with ammonia has to be written and the name of the carboxylic acid salt formed should be named.
Concept introduction:
Carboxylic acids contain a carbonyl attached to a hydroxyl group as shown below,

Nomenclature of carboxylic acid:
- Find the Parent hydrocarbon chain.
- Carboxyl carbon must be numbered first.
- Replace the –e in the alkane name with –oic acid.
Carboxylic acid reacts with ammonia gives neutral carboxylic salt.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
OWLv2 with MindTap Reader, 1 term (6 months) Printed Access Card for Brown/Iverson/Anslyn/Foote's Organic Chemistry, 8th Edition
- Write an equation for the reaction of chloroacetic acid (Ka=1.5103) with trimethylamine (Kb=5.9105) . Calculate the equilibrium constant for the reaction. If 0.10 M solutions of these two species are mixed, what will be their concentrations at equilibrium?arrow_forwardThe amount of tartaric acid is responsible for the tartness of wine and controls the acidity of the wine. Tartaric acid also plays a very significant role in the overall taste, feel and color of a wine. Tartaric acid is a diprotic organic acid The chemical formula for tartaric acid is C4H6O6 and its structural formula is HO2CCH(OH)CH(OH)CO2H. A 50.00 mL sample of a white dinner wine required 21.48 mL of 0.03776 M NaOH to achieve a faint pink color. Express the acidity of the wine in terms of grams of tartaric acid, H2C4H4O6 (M. M. = 150.10) per 100 mL of wine. Assume that the two acidic hydrogens are titrated at the end point. MM H2C4H4O6 = 150.10 MM NaOH = 40.00 Below is the balanced chemical equation for this titration.arrow_forwardWrite the chemical equation for the acid dissociation of acetaminophen, C8H9O2N. Write the Ka expression for the acid dissociation of acetaminophen.arrow_forward
- What is the solubility, the pH, and reaction of carboxylic acids?arrow_forwardShow how methanol (CH3OH) can serve as both an acid and a base. Write an equation for the reaction of methanol with sulfuric acid.arrow_forwardWrite the products of the reaction of diphenhydramine (a base) with the acid HCl shown below. Are the reactants or products more soluble in water? Briefly explain.arrow_forward
- H3C—N̈H2 reacts with BF3 in a combination reaction, forming H3C—NH2—BF3 with a covalent bond between N and B. What is the role of H3C—N̈H2 in this reaction? a.Brønsted-Lowry acid b.Lewis base c.Brønsted-Lowry base d.Lewis acidarrow_forwardSalicylic acid preparation and propertiesarrow_forwardWrite the chemical equation between acetic acid and sodium hydroxide.arrow_forward
- Draw the Lewis structures for chloroacetic acid and for acetic acid. Use structural arguments to explain why acetic acid is a weaker acid then chloroacetic acid. Be sure to refer to the stability of the ion that would be formed after dissociation of the acid.arrow_forwardwrite the esterification reaction of lauric acid with ethanolarrow_forwardSynthesis of Benzoic Acid Is the reaction between sodium benzoate and hydrochloric acid exothermic or endothermic? What type of reaction is this? What are the products? Why is ice cold water rather than room temperature or warm water used to wash the remainder of the benzoic acid out of the beaker?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





